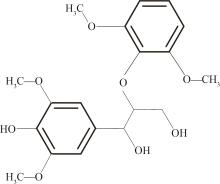
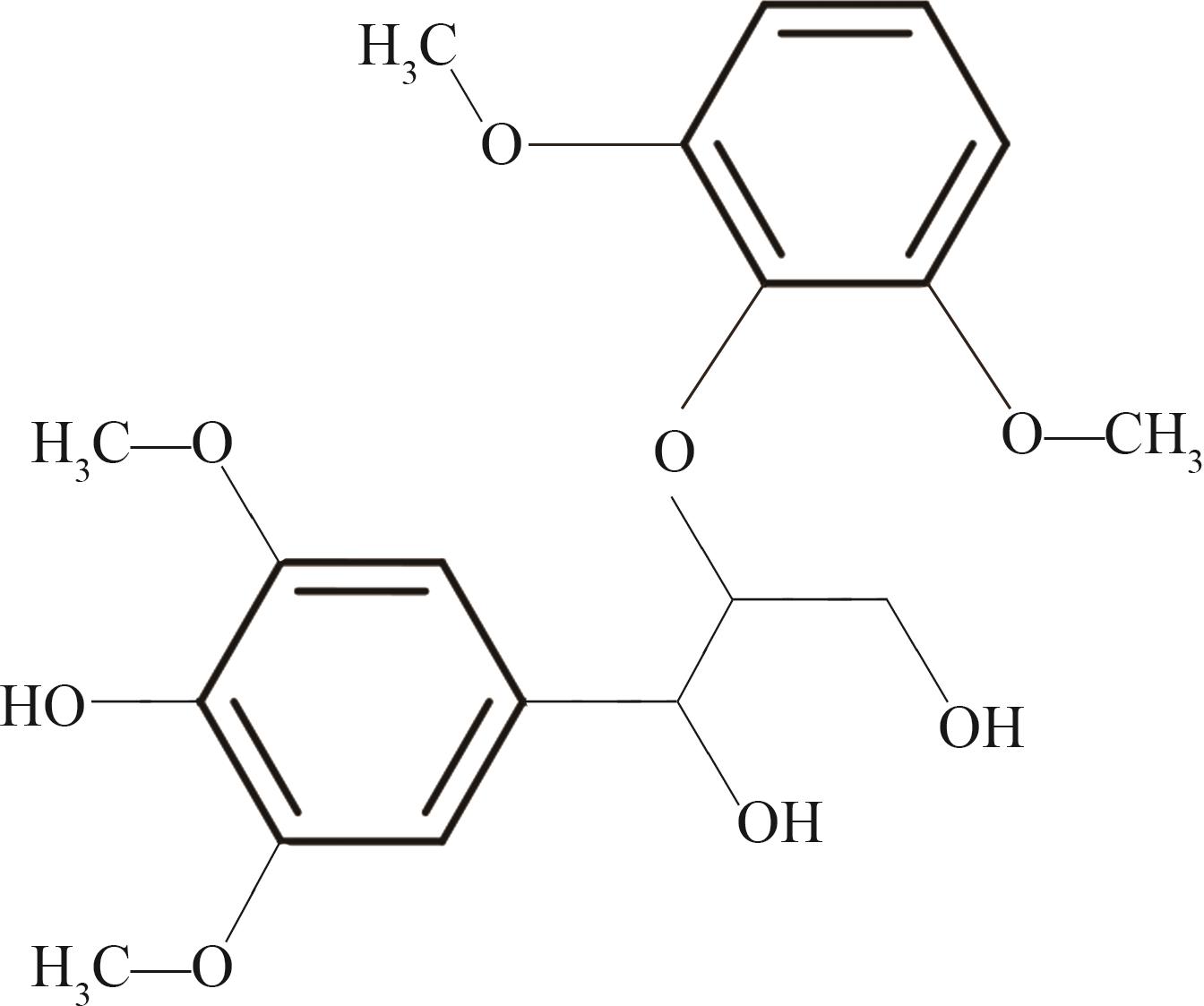
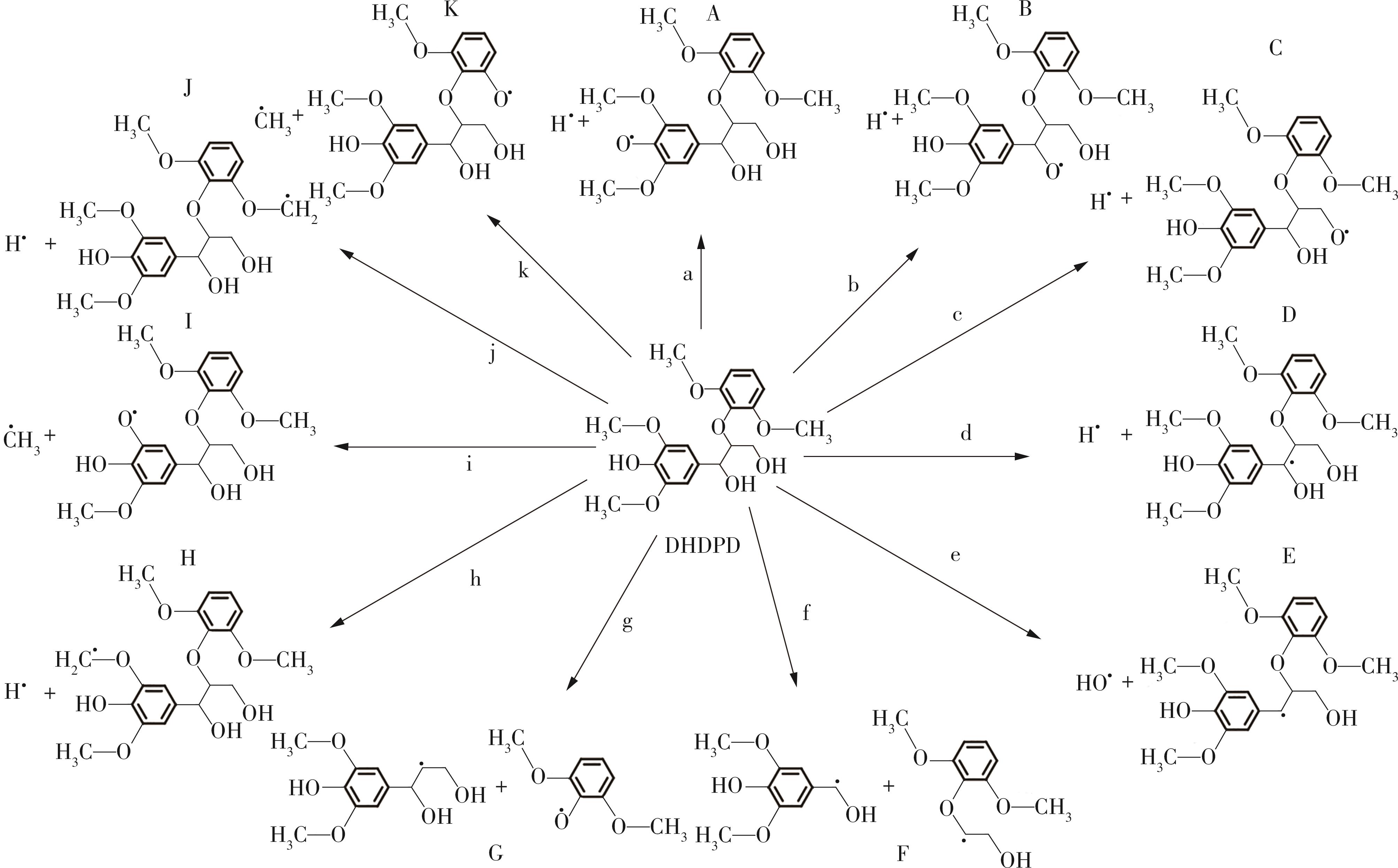
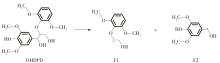
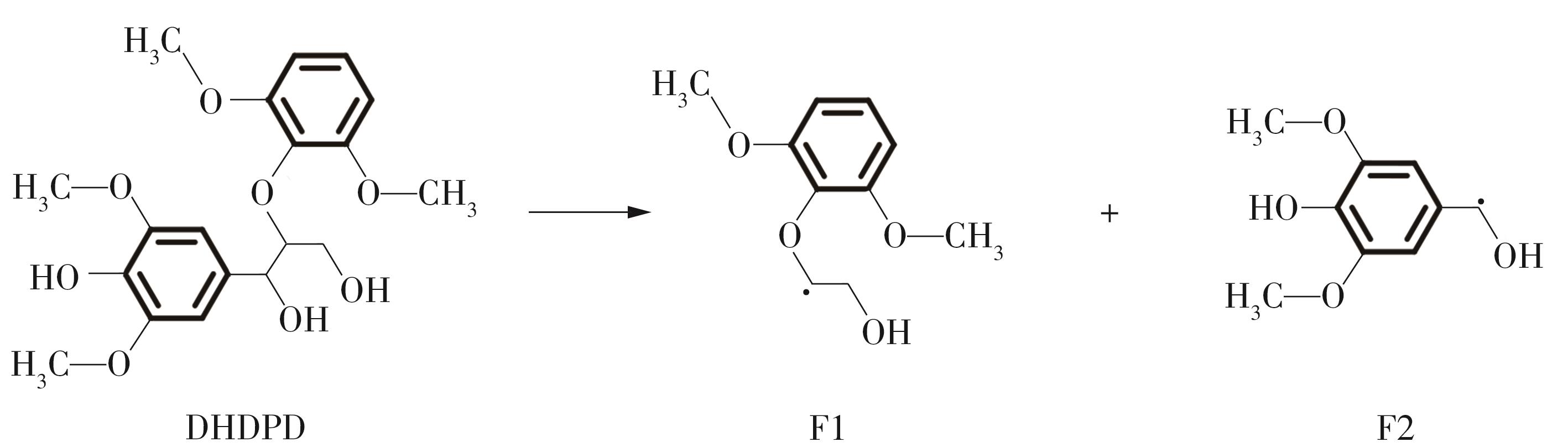
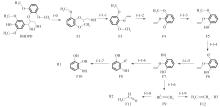
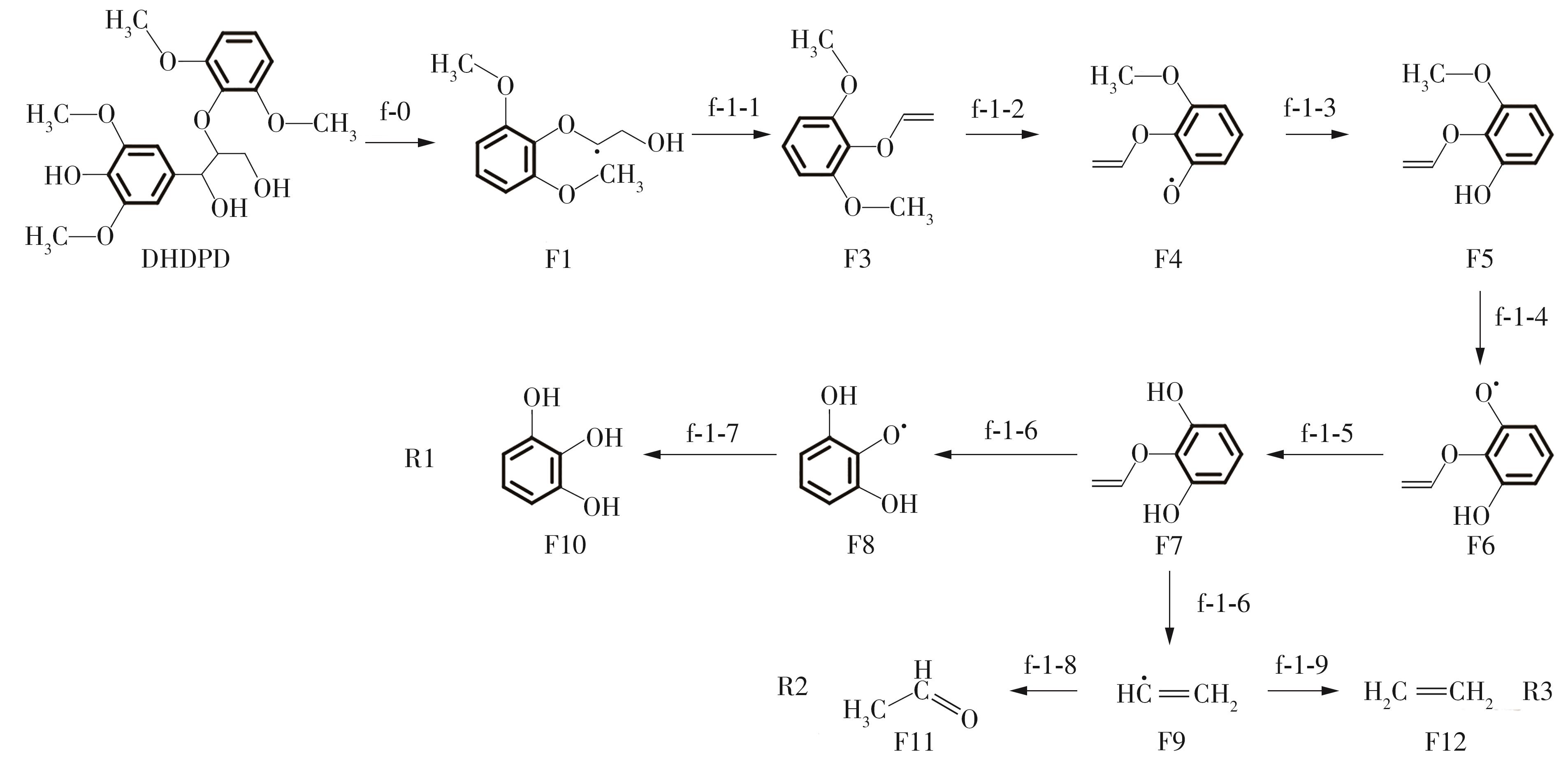
Journal of South China University of Technology(Natural Science Edition) ›› 2023, Vol. 51 ›› Issue (12): 107-117.doi: 10.12141/j.issn.1000-565X.220555
Special Issue: 2023年能源、动力与电气工程
• Energy, Power & Electrical Engineering • Previous Articles Next Articles
Kinetic Mechanism of Pyrolysis of Lilac Lignin Dimer Memes
LOU Bo LI Senhao LU Song ZHOU Daheng
- School of Electric Power Engineering/Guangdong Province Key Laboratory of Energy Efficient and Clean Utilization,South China University of Technology,Guangzhou 510640,Guangdong,China
-
Received:2022-08-26Online:2023-12-25Published:2022-12-06 -
About author:楼波(1965-),男,博士, 副教授,主要从事燃烧理论与节能技术研究。E-mall: loubo@scut.edu.cn -
Supported by:the Natural Science Foundation of Guangdong Province(2021B1515020071)
CLC Number:
Cite this article
LOU Bo, LI Senhao, LU Song, et al. Kinetic Mechanism of Pyrolysis of Lilac Lignin Dimer Memes[J]. Journal of South China University of Technology(Natural Science Edition), 2023, 51(12): 107-117.
share this article
Table 2
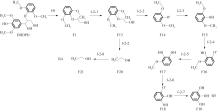
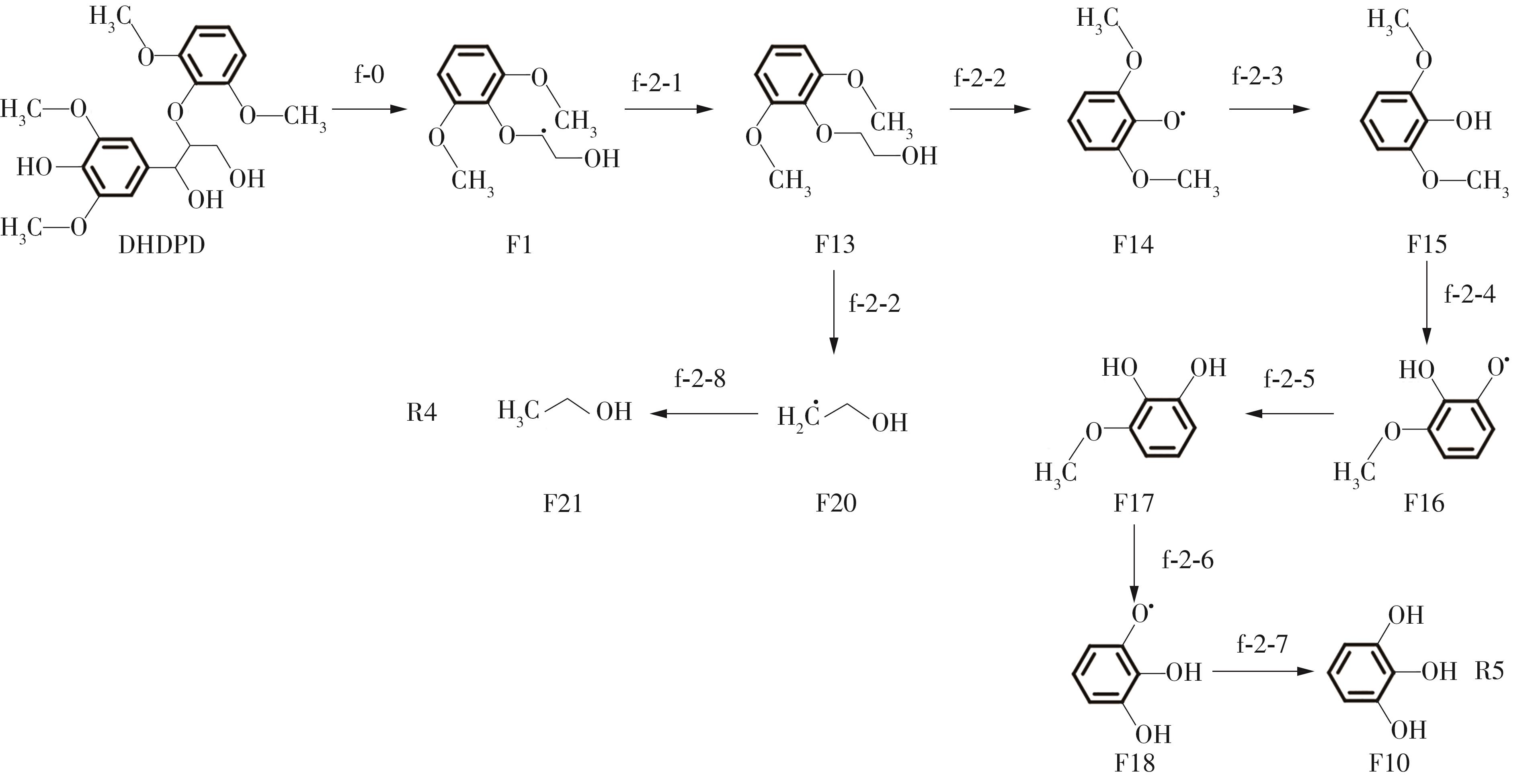
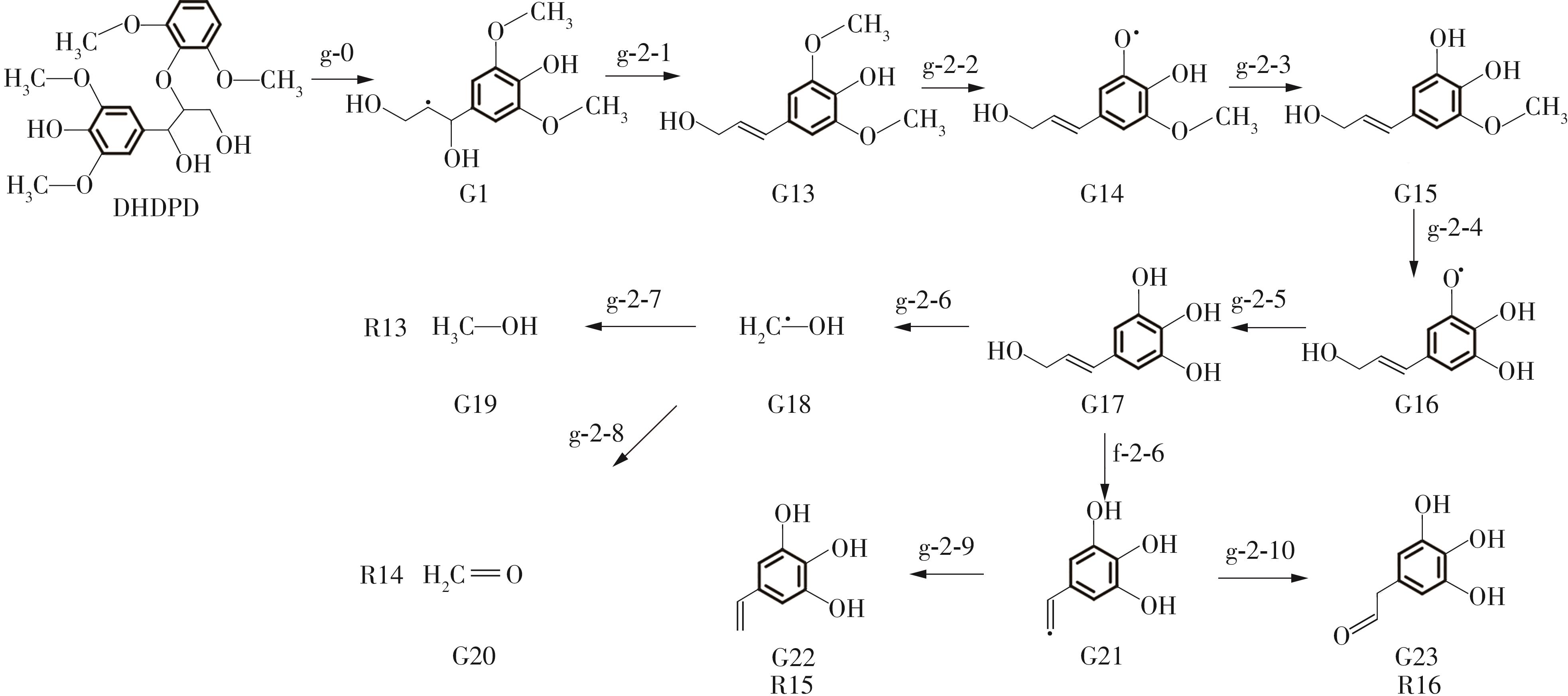
Enthalpy change of decomposition reaction at each step of f path"
| 反应 | ΔH/(kJ·mol-1) | 反应 | ΔH/(kJ·mol-1) | ||
|---|---|---|---|---|---|
| f-0 | 256.47 | f-3-3 | -458.86 | ||
| f-1-1 | 134.34 | f-3-4 | 333.36 | ||
| f-1-2 | 340.35 | f-3-5 | -325.64 | ||
| f-1-3 | -339.43 | f-3-6 | 473.09 | ||
| f-1-5 | 327.66 | f-3-7 | -35.14 | ||
| f-1-6 | -316.98 | f-3-8 | 109.08 | ||
| f-1-7 | -445.51 | f-3-9 | -356.59 | ||
| f-1-8 | -406.63 | f-3-10 | 80.46 | ||
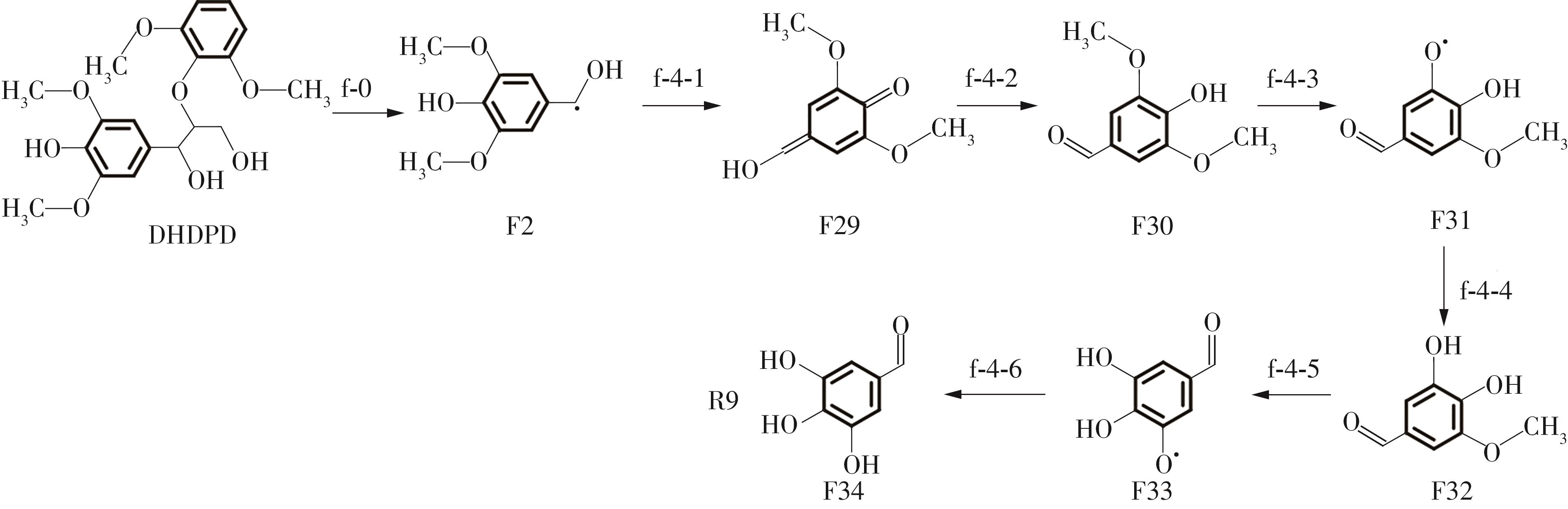
| f-1-9 | -531.65 | f-4-1 | 150.92 | ||
| f-2-1 | -252.27 | f-4-2 | 33.93 | ||
| f-2-2 | 320.01 | f-4-3 | 297.31 | ||
| f-2-3 | -315.55 | f-4-4 | -426.72 | ||
| f-2-4 | 302.96 | f-4-5 | 334.66 | ||
| f-2-5 | -259.1 | f-4-6 | -311.56 | ||
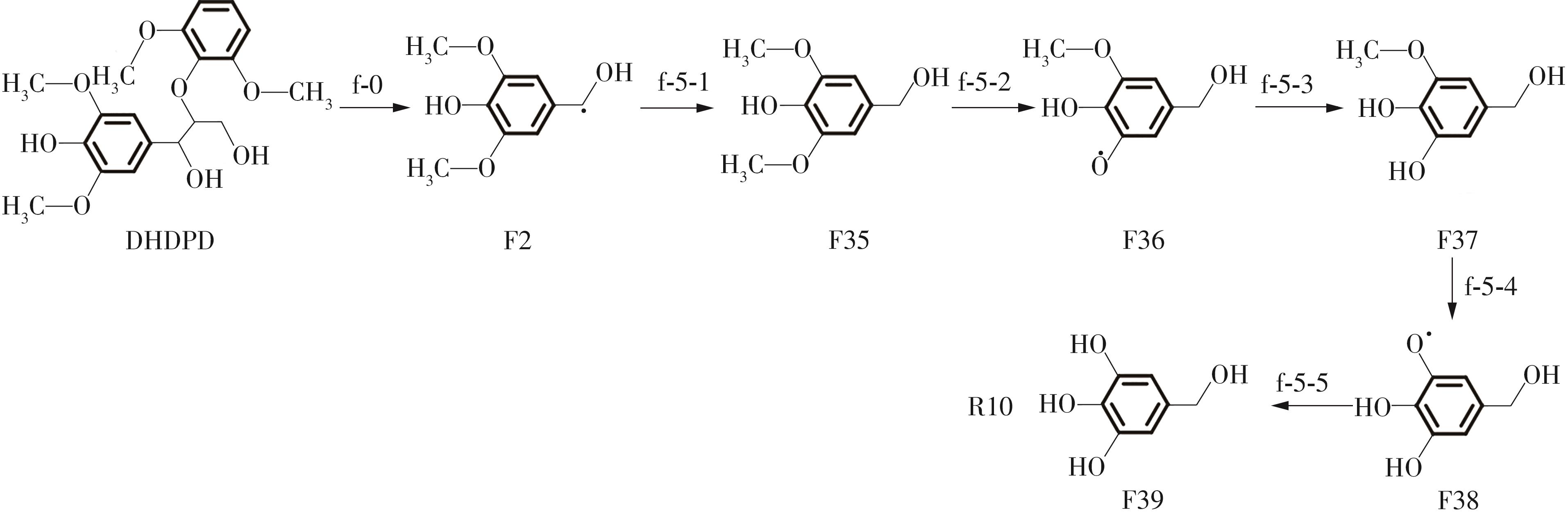
| f-2-6 | 329.05 | f-5-1 | -424.95 | ||
| f-2-7 | -300.81 | f-5-2 | 304.36 | ||
| f-2-8 | -383.86 | f-5-3 | -400.68 | ||
| f-3-1 | -252.27 | f-5-4 | 332.45 | ||
| f-3-2 | 348.43 | f-5-5 | -287.09 | ||
Table 3
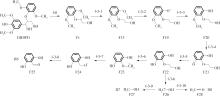
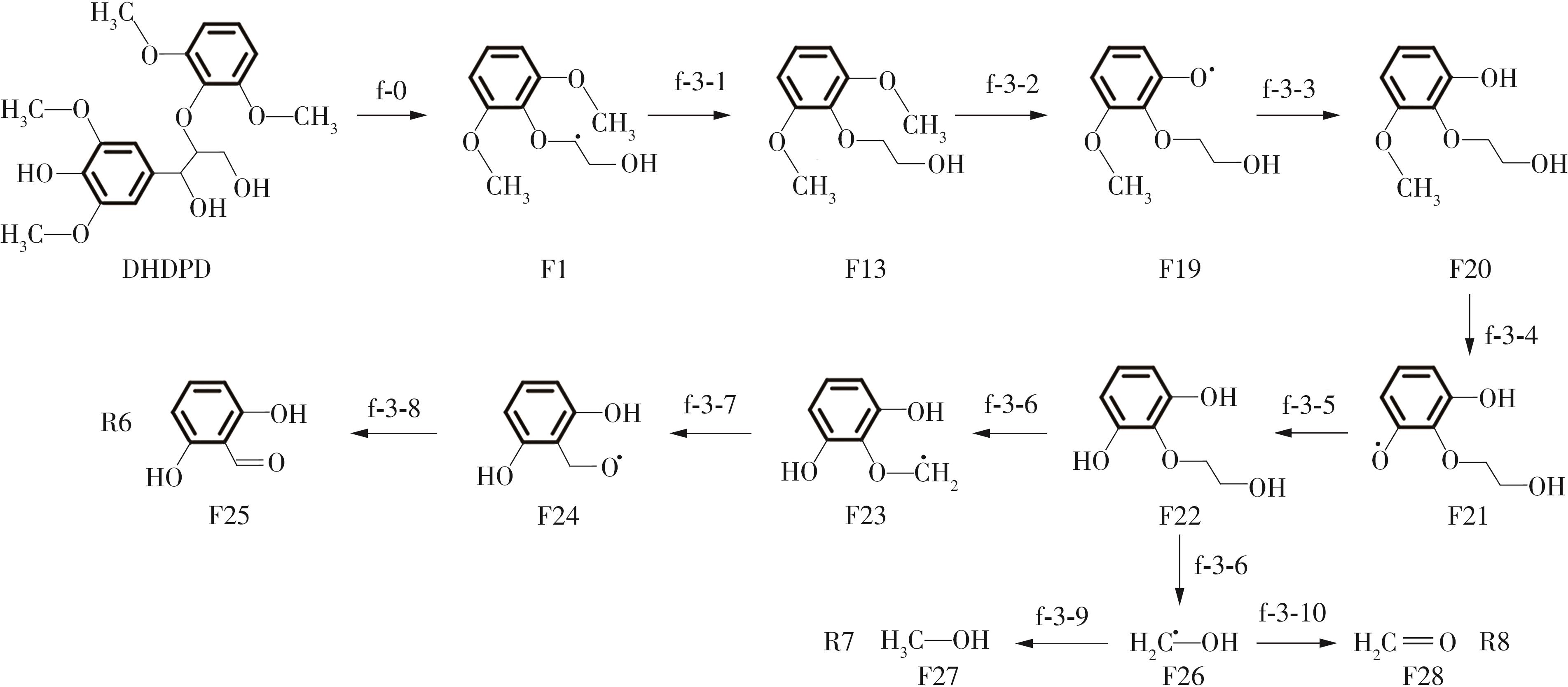
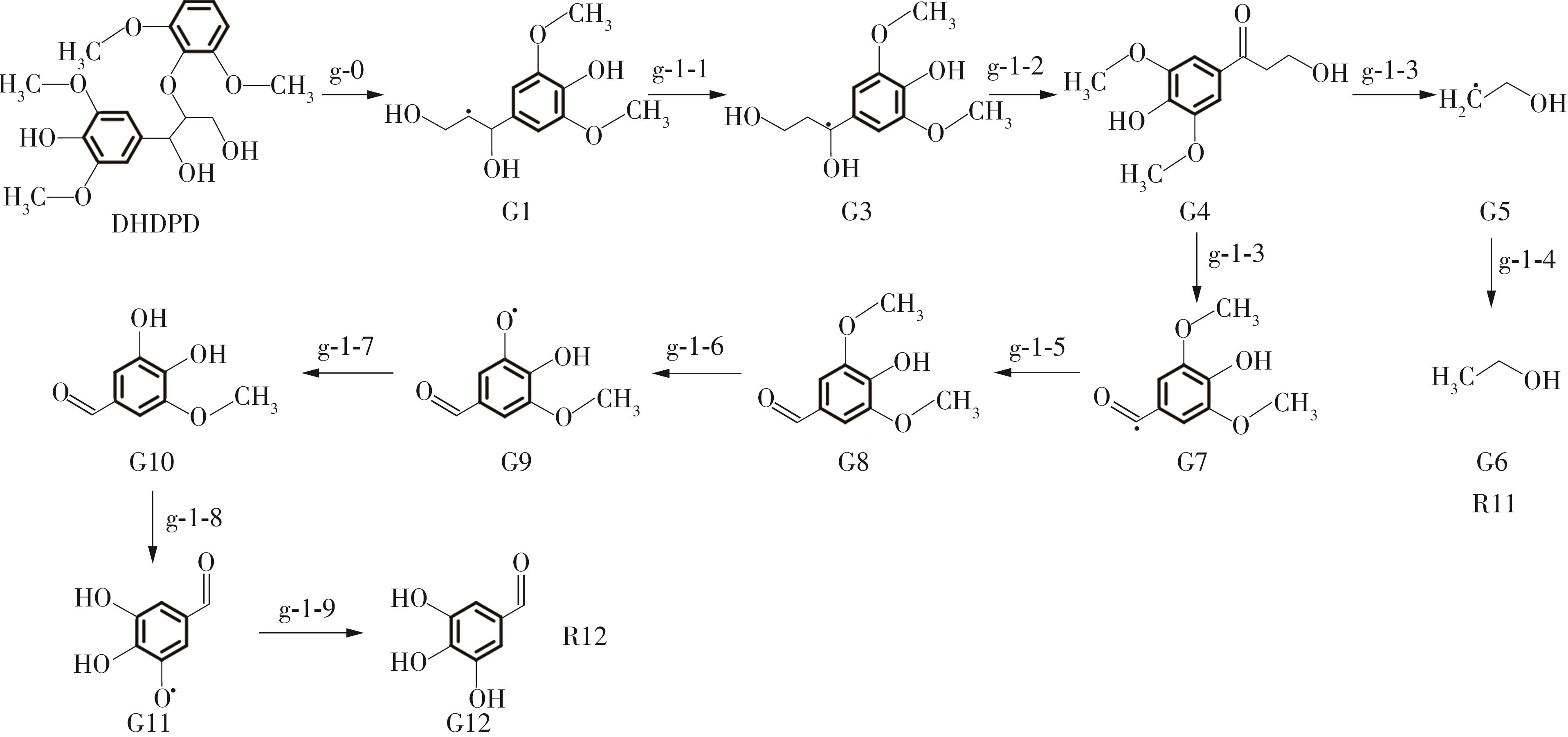
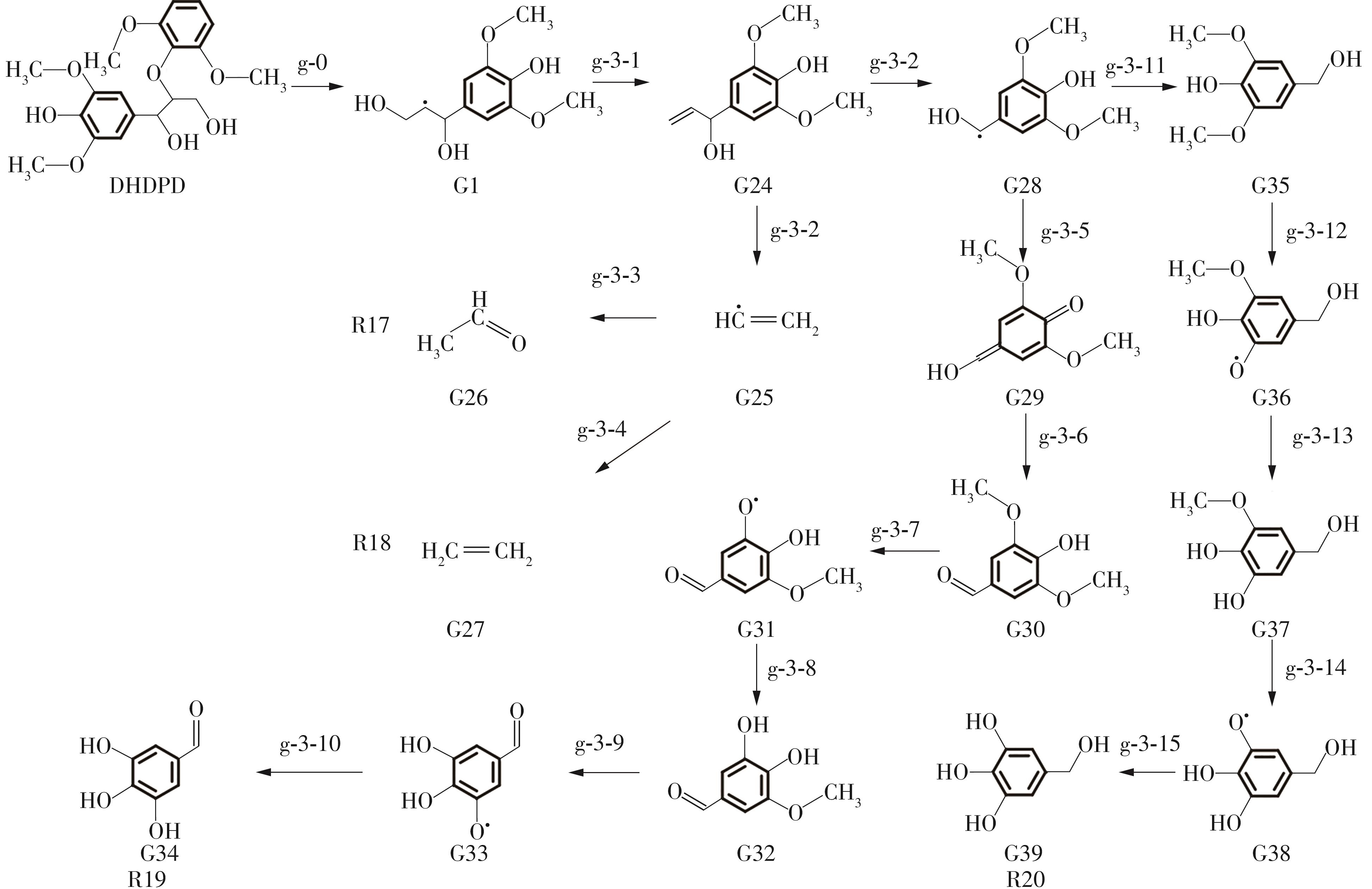
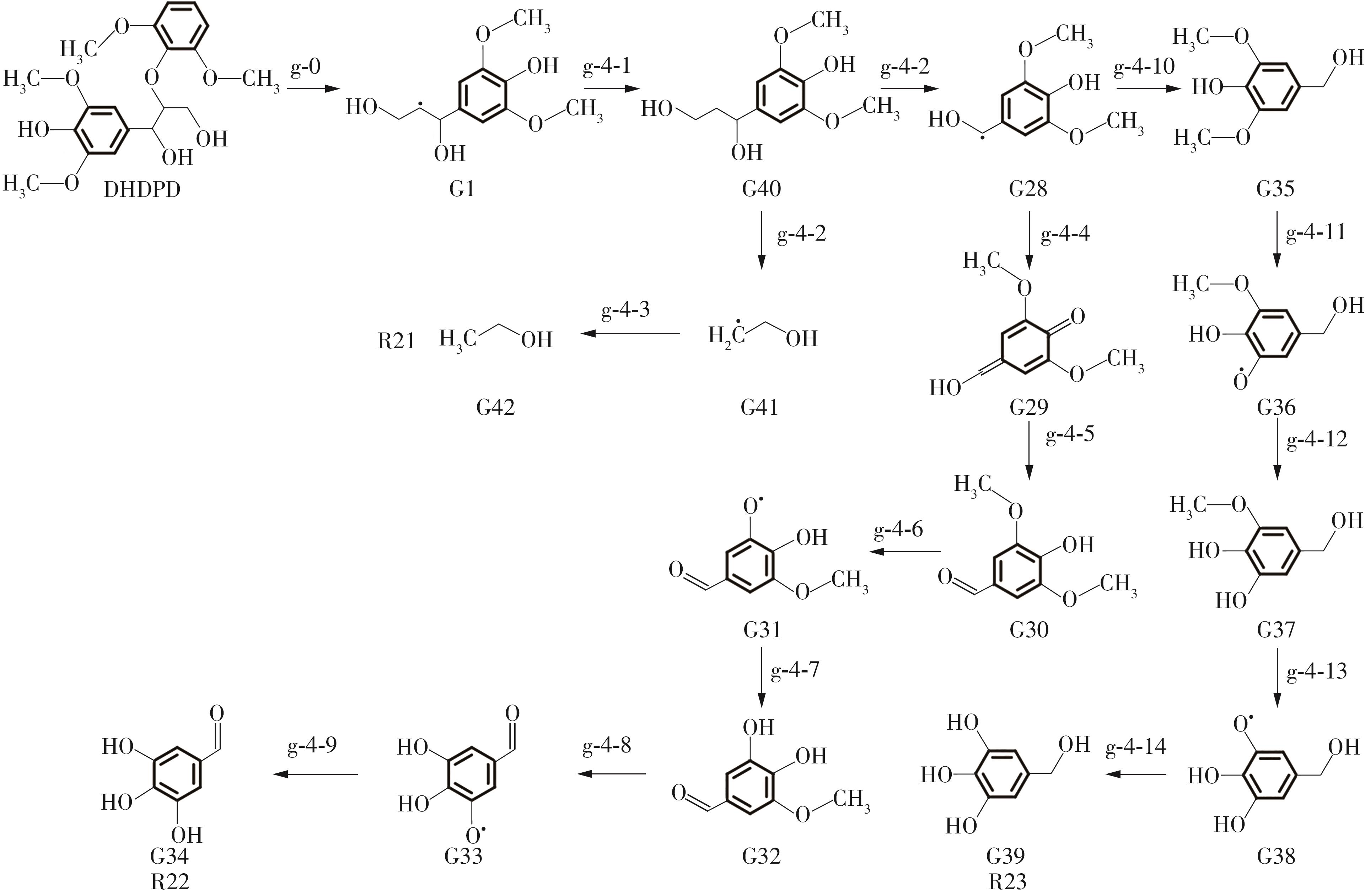
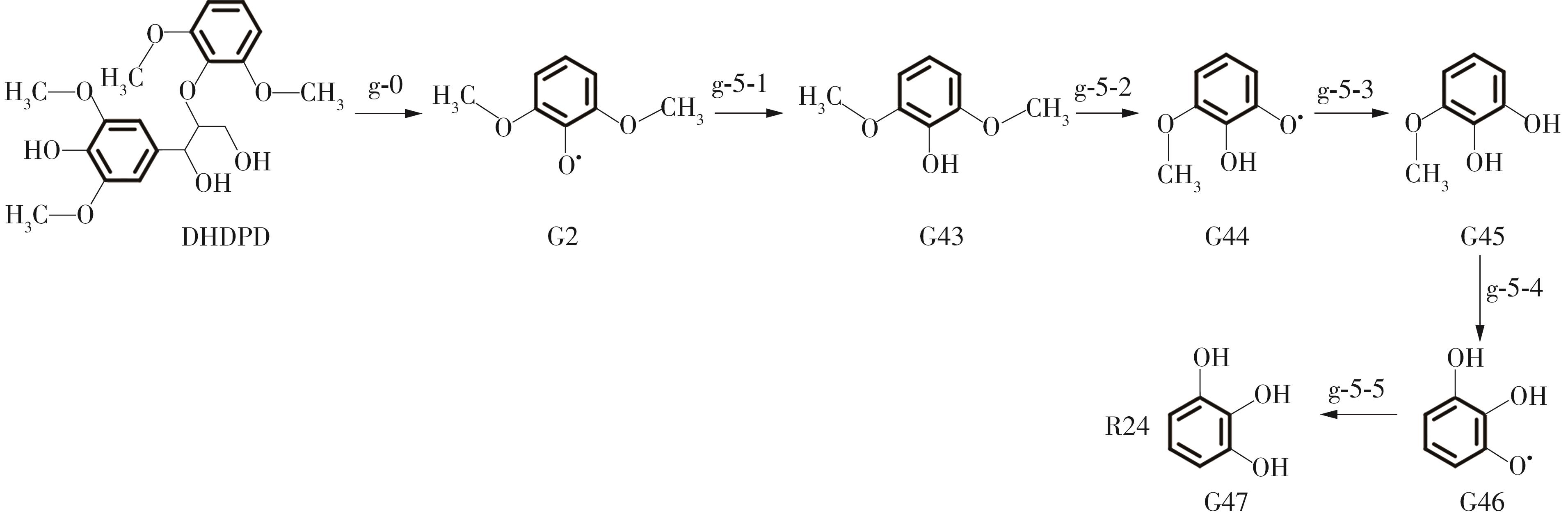
Enthalpy change of decomposition reaction at each step of g path"
| 反应 | ΔH/(kJ·mol-1) | 反应 | ΔH/(kJ·mol-1) | ||
|---|---|---|---|---|---|
| g-0 | 185.18 | g-3-8 | -426.72 | ||
| g-1-1 | -31.28 | g-3-9 | 334.66 | ||
| g-1-2 | 54.26 | g-3-10 | -311.56 | ||
| g-1-3 | 294.10 | g-3-11 | -424.95 | ||
| g-1-4 | -383.86 | g-3-12 | 304.36 | ||
| g-1-5 | -332.24 | g-3-13 | -400.68 | ||
| g-1-6 | 217.77 | g-3-14 | 332.45 | ||
| g-1-7 | -288.98 | g-3-15 | -287.09 | ||
| g-1-8 | 315.18 | g-4-1 | -560.56 | ||
| g-1-9 | -428.92 | g-4-2 | 369.95 | ||
| g-2-1 | 155.58 | g-4-3 | -383.86 | ||
| g-2-2 | 203.23 | g-4-4 | 150.92 | ||
| g-2-3 | -347.45 | g-4-5 | 33.93 | ||
| g-2-4 | 320.54 | g-4-6 | 297.31 | ||
| g-2-5 | -304.32 | g-4-7 | -426.72 | ||
| g-2-6 | 367.71 | g-4-8 | 334.66 | ||
| g-2-7 | -444.06 | g-4-9 | -311.56 | ||
| g-2-8 | -210.59 | g-4-10 | -409.90 | ||
| g-2-9 | 80.46 | g-4-11 | 304.36 | ||
| g-2-10 | -356.59 | g-4-12 | -400.68 | ||
| g-3-1 | 212.04 | g-4-13 | 332.45 | ||
| g-3-2 | 408.94 | g-4-14 | -287.09 | ||
| g-3-3 | -531.65 | g-5-1 | 311.20 | ||
| g-3-4 | -406.63 | g-5-2 | 183.30 | ||
| g-3-5 | 150.92 | g-5-3 | -247.30 | ||
| g-3-6 | 33.93 | g-5-4 | 165.30 | ||
| g-3-7 | 297.31 | g-5-5 | -252.00 | ||
| 1 | DEMIRBAS A .Biomass resource facilities and biomass conversion processing for fuels and chemicals[J].Energy Conversion & Management,2001,42(11):1357-1378. |
| 2 | ABDULLAH H, WU H .Biochar as a fuel:1.properties and grindability of biochars produced from the pyrolysis of mallee wood under slow-heating conditions[J].Energy & Fuels,2009,23(8):4174-4181. |
| 3 | LIN J C M .Development of a high yield and low cycle time biomass char production system[J].Fuel Processing Technology,2006,87(6):487-495. |
| 4 | 胡正文,张建良,左海滨,等 .生物质能辅助炼铁状况及前景[C]∥2012年全国炼铁生产技术会议暨炼铁学术年会文集(下).无锡:中国金属学会,2012:671-677. |
| 5 | 李文涛,柴宝华,王美净,等 .不同生活垃圾组分热解炭化特性与热解焦傅里叶红外光谱表征[J].新能源进展,2020,8(1):22-27. |
| LI Wen-tao, CHAI Bao-hua, WANG Mei-jing,et al .Pyrolysis carbonization and char FTIR characterization study on different components of municipal solid waste [J].New Energy Progress,2020,8(1):22-27. | |
| 6 | 付跃进,杨昇,王方骏,等 .核桃壳木质素的结构研究[J].林业工程学报,2018,3(3):88-94. |
| FU Yue-jin, YANG Sheng, WANG Fang-jun,et al .Structural characterization of lignin from walnut shell [J].Journal of Forestry Engineering,2018,3(3):88-94. | |
| 7 | 王惠,杨海峰,翟高红,等 .碳源甲基苯热裂解机理的密度泛函动力学研究[J].化学学报,2001,59(1):17-21. |
| WANG Hui, YANG Hai-feng, ZHAI Gao-hong,et al .DFT kinetic study of the pyrolysis mechanism of toluene used for carbon matrix [J].Journal of Chemistry,2001,59(1):17-21. | |
| 8 | 王惠,罗瑞盈,杨延清,等 .2,4-二甲基卤代苯热解机理的理论研究[J].西北大学学报(自然科学版),2001,31(1):33-36. |
| WANG Hui, LUO Rui-ying, YANG Yan-qing,et al .A theoretical study on pyrolysis mechanism of a series of compounds 2,4-dimethylhalobenzene[J].Journal of Northwest University (Natural Science Edition),2001,31(1):33-36. | |
| 9 | 翟高红,朱卡克,王惠,等 .碳前驱体热解机理的量子化学理论研究——几何结构、反应焓变、化学键和热反应活性[J].材料科学与工程,2000,18(4):10-15. |
| ZHAI Gao-hong, ZHU Ka-ke, WANG Hui,et al .Quantum chemical theoretical research of pyrolysis mechanism of carbon precursors-geometry,enthalpy change,chemical bond and thermoreactive activity[J].Materials Science and Engineering,2000,18(4):10-15. | |
| 10 | 王惠,翟高红,冉新权,等 .碳材料用碳源化合物热解机理的理论研究[J].无机化学学报,2000,16(6):879-887. |
| WANG Hui, ZHAI Gao-hong, RAN Xin-quan,et al .Theoretical research of the pyrolysis mechanism of carbon matrix precursor used for carbon materials[J].Journal of Inorganic Chemistry,2000,16(6):879-887. | |
| 11 | 乔占平,王惠,赵文立,等 .甲基苯热裂解机理的理论研究[J].燃料化学学报,2002,30(1):49-53. |
| QIAO Zhan-ping, WANG Hui, ZHAO Wen-li,et al .Theoretical study on pyrolysis mechanism of toluene [J].Journal of Fuel Chemistry,2002,30(1):49-53. | |
| 12 | 王华静,赵岩,王晨,等 .木质素二聚体模型物裂解历程的理论研究[J].化学学报,2009,67(9):893-900. |
| WANG Hua-jing, ZHAO Yan, WANG Chen,et al .Theoretical study on the pyrolysis process of lignin dimer model[J].Acta Chimica Sinica,2009,67(9):893-900. | |
| 13 | BRITT P F, BUCHANAN A C, COONEY M J,et al .Flash vacuum pyrolysis of methoxy-substituted lignin model compounds[J].Journal of Organic Chemistry,2000,65(5):1376-1389. |
| 14 | CHENG Y H, ZHAO X, SONG K S,et al .Remote substituent effects on bond dissociation energies of para-substituted aromatic silanes[J].Journal of Organic Chemistry,2002,67(19):6638-6645. |
| 15 | SONG K S, LIU L, GUO Q X. Remote substituent effects on N-X(X = H,F,Cl,CH3,Li)bond dissociation energies in para-substituted anilines[J].Journal of Organic Chemistry,2003,68(2):262-266. |
| 16 | 刘江燕 .木质素及其模型物在不同热化学环境下的解构[D].广州:华南理工大学,2010. |
| 17 | KAWAMOTO H, HORIGOSHI S, SAKA S .Pyrolysis reactions of various lignin model dimers[J].Journal of Wood Science,2007,53(2),168-174. |
| 18 | LIU C, ZHANG Y, HUANG X .Study of guaiacol pyrolysis mechanism based on density function theory [J].Fuel Processing Technology,2014,123:159-165. |
| 19 | ASMADI M, KAWAMOTO H, SAKA S .Thermal reactions of guaiacol and syringol as lignin model aromatic nuclei[J].Journal of Analytical and Applied Pyrolysis,2011,92(1):88-98. |
| [1] | ZHENG Zhi, GUO Naisheng, YOU Zhanping. Molecular Simulation of Interaction Behavior Between Asphalt Components and Waste Wood Oil [J]. Journal of South China University of Technology(Natural Science Edition), 2024, 52(12): 79-86. |
| [2] | TANG Yuting, CHEN Xiaobin, MA Xiaoqian. Experiment Study of Capturing CO2 from Pyrolysis Gases by Zr-Mg Modified Sorbents [J]. Journal of South China University of Technology(Natural Science Edition), 2022, 50(7): 118-125. |
| [3] | XU Ning, WANG Hainian, CHEN Yu, et al. Research on the Compatibility of Bio-Asphalt Based on Molecular Dynamics [J]. Journal of South China University of Technology(Natural Science Edition), 2022, 50(5): 65-72. |
| [4] | ZHAO Jin, QIN Yangjun, LIU Chang, et al. Research on Graphene-based Heat Transfer in Nanochannel by Using Non-equilibrium Molecular Dynamics [J]. Journal of South China University of Technology(Natural Science Edition), 2022, 50(3): 140-146. |
| [5] | LIAO Yanfen, ZHANG Manyu, CHEN Shunkai, et al. Study on MILD Combustion of MSW Pyrolysis Gas Based on FLUENT [J]. Journal of South China University of Technology (Natural Science Edition), 2021, 49(2): 9-16. |
| [6] | LIAO Yanfen, CHEN Shunkai, ZHANG Manyu, et al. Numerical Analysis of MILD Combustion of Organic Solid Waste Pyrolysis Gas [J]. Journal of South China University of Technology (Natural Science Edition), 2020, 48(7): 28-35,46. |
| [7] | HOU Yi ZHANG Xiao LIU Chao LI Youming. Dissolution Mechanism of Wheat Straw Alkaline Lignin in Alkaline Ionic Liquid [Bmim][OH] [J]. Journal of South China University of Technology(Natural Science Edition), 2019, 47(5): 18-24,31. |
| [8] | ZHAO Yuanyuan WU Shubin LIAO Yanfen . Depolymerization Property of Alkali Lignin in Solvent/Water System [J]. Journal of South China University of Technology(Natural Science Edition), 2019, 47(5): 32-38. |
| [9] | PANG Yu-xia CAI Meng LOU Hong-ming . Preparation and Pb2+ Removal Capacity of Phosphorylated Lignin [J]. Journal of South China University of Technology (Natural Science Edition), 2017, 45(6): 96-102. |
| [10] | CHANG Jie KASSANOV Bekbolat WANG Ju LIU Jun FU Yan. Synthesis of Phenol-Furfural Resins Using Hydrogenolysis Products of Lignin Regenerated via Ionic Liquid Pretreatment [J]. Journal of South China University of Technology (Natural Science Edition), 2017, 45(6): 89-95. |
| [11] | ZHAN Jia-ming YAO Xiao-hu LI Wang-hui. A Probe into Mechanical Properties of Graphene/ SiC Composites Based on Molecular Dynamics [J]. Journal of South China University of Technology (Natural Science Edition), 2017, 45(5): 135-140. |
| [12] | LIU Hao SUN Jian-liang Puneet K.SINGH YANG Wen-ping FU Shi-yu . Molecular Docking of Myceliophthora thermophila Laccase with Lignin Model Compounds [J]. Journal of South China University of Technology (Natural Science Edition), 2017, 45(11): 112-119. |
| [13] | CHANG Jie LIU Jun GUO Shu-jun WANG Xi FU Yan. Investigation into Selective Separation of Lignin in Novel Deep Eutectic Solvent [J]. Journal of South China University of Technology (Natural Science Edition), 2016, 44(6): 14-20,26. |
| [14] | QIU Xue-qing CHEN Jian-hao CAI Zhen-he ZHOU Ming-song. Color Masking and Mechanical Properties of Lignin/High-Density Polyethylene Composites [J]. Journal of South China University of Technology (Natural Science Edition), 2016, 44(6): 1-8. |
| [15] | LI You-ming CHEN Jing-tian LEI Li-rong. Effect and Kinetics Analysis of Eugenol Degradation by Ozone [J]. Journal of South China University of Technology (Natural Science Edition), 2016, 44(6): 90-97. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||