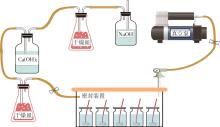
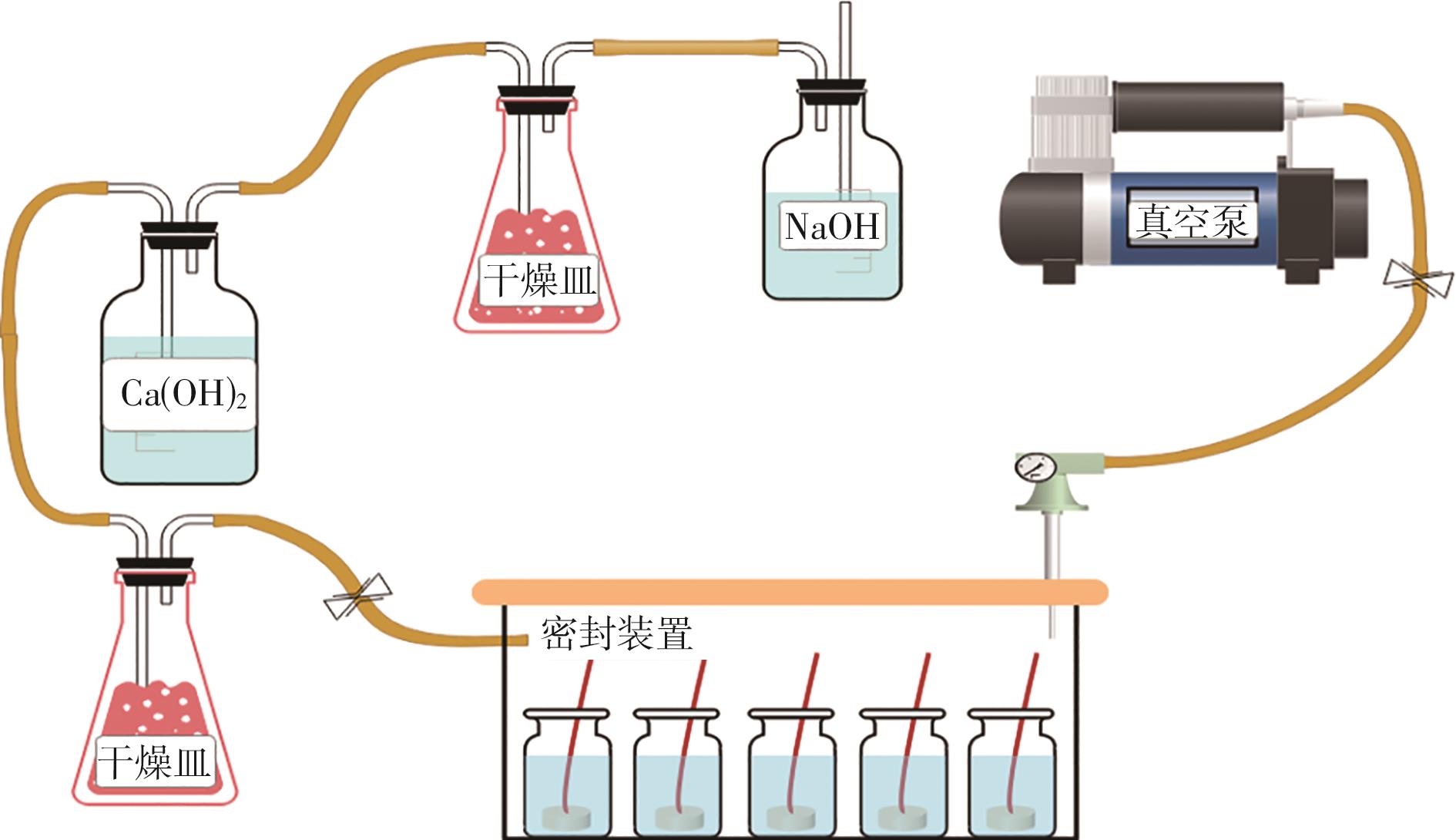
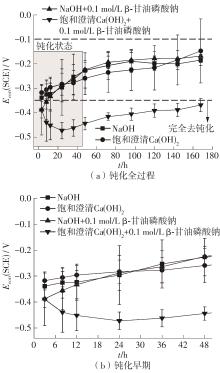
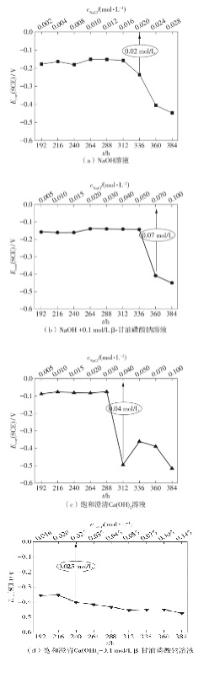
Journal of South China University of Technology(Natural Science Edition) ›› 2024, Vol. 52 ›› Issue (3): 28-40.doi: 10.12141/j.issn.1000-565X.230030
• Materials Science & Technology • Previous Articles Next Articles
Study on Sodium β-glycerophosphate in Concrete Affects the Inhibited Behavior of Steel Bar
WANG Xiaoxian1 LIU Jiaping1 MU Song2 JIN Ming1 MA Qi2
- 1.School of Materials Science and Engineering,Southeast University,Nanjing 211189,Jiangsu,China
2.National Key Laboratory of High Performance Civil Engineering Materials,Jiangsu Building Science Research Institute Co. ,Ltd. ,Nanjing 210008,Jiangsu,China
-
Received:2023-01-05Online:2024-03-25Published:2024-03-05 -
Contact:刘加平(1967-),男,教授、博士生导师,主要从事钢筋混凝土耐久性研究。 E-mail:liujiaping@cmjgk.cn -
About author:王潇舷(1992-),女,博士生,主要从事钢筋混凝土耐久性研究。E-mail:xiaoxian_wang@foxmail.com -
Supported by:the National Key Research and Development Project of China(2021YFF0500802,2021YFF0500803); the Southeast University Outstanding Doctoral Dissertation Cultivation Fund(YBPY21)
CLC Number:
Cite this article
WANG Xiaoxian, LIU Jiaping, MU Song, et al. Study on Sodium β-glycerophosphate in Concrete Affects the Inhibited Behavior of Steel Bar[J]. Journal of South China University of Technology(Natural Science Edition), 2024, 52(3): 28-40.
share this article
Table 2
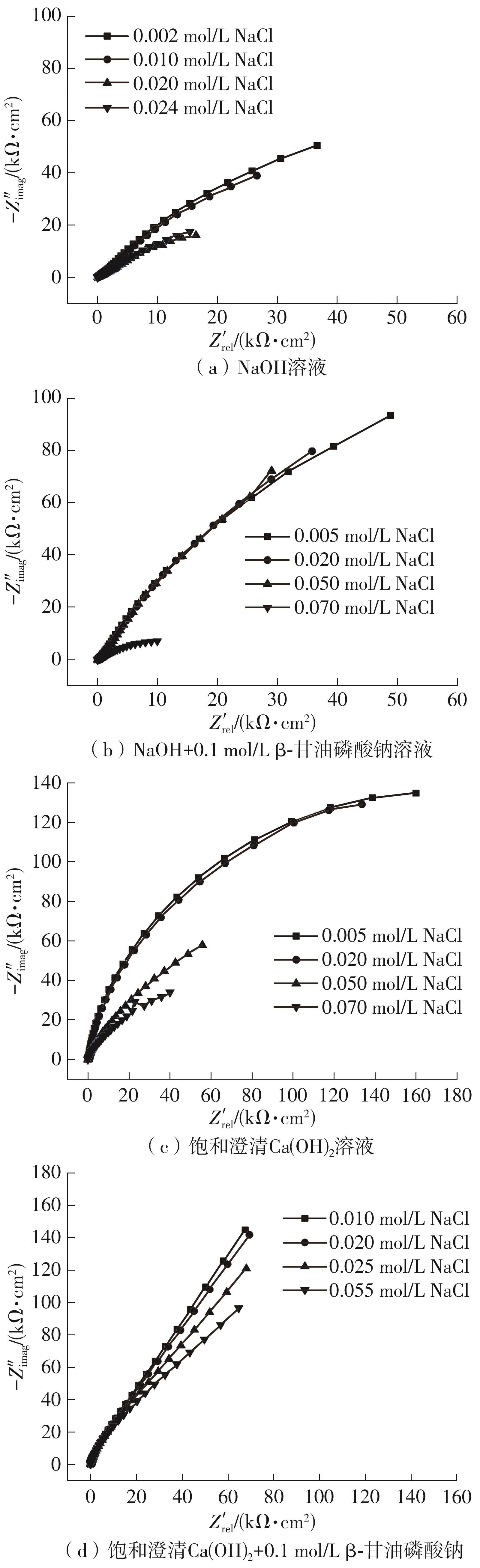
Cumulative Cl- regimes in different solutions"
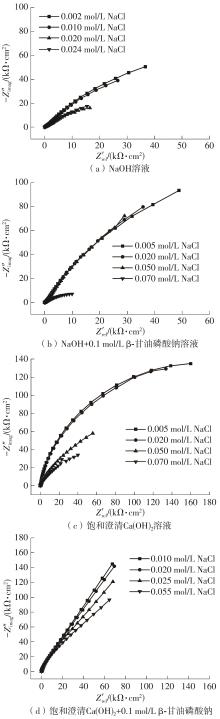
| 溶液类型 | NaCl添加增量/(mol·L-1) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 开始 | 1次 | 2次 | 3次 | 4次 | 5次 | 6次 | 7次 | 8次 | |
| NaOH | 0.002 | 0.004 | 0.008 | 0.010 | 0.012 | 0.016 | 0.020 | 0.024 | 0.028 |
| NaOH+0.1 mol/L β-甘油磷酸钠 | 0.005 | 0.010 | 0.015 | 0.020 | 0.030 | 0.040 | 0.050 | 0.070 | 0.100 |
| 饱和澄清Ca(OH)2 | 0.005 | 0.010 | 0.015 | 0.020 | 0.030 | 0.040 | 0.050 | 0.070 | 0.100 |
| 饱和澄清Ca(OH)2+0.1 mol/L β-甘油磷酸钠 | 0.010 | 0.020 | 0.025 | 0.035 | 0.045 | 0.055 | 0.075 | 0.105 | 0.145 |
Table 3
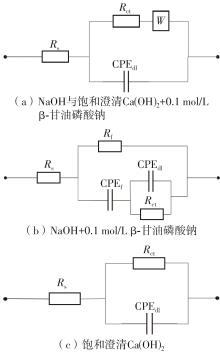
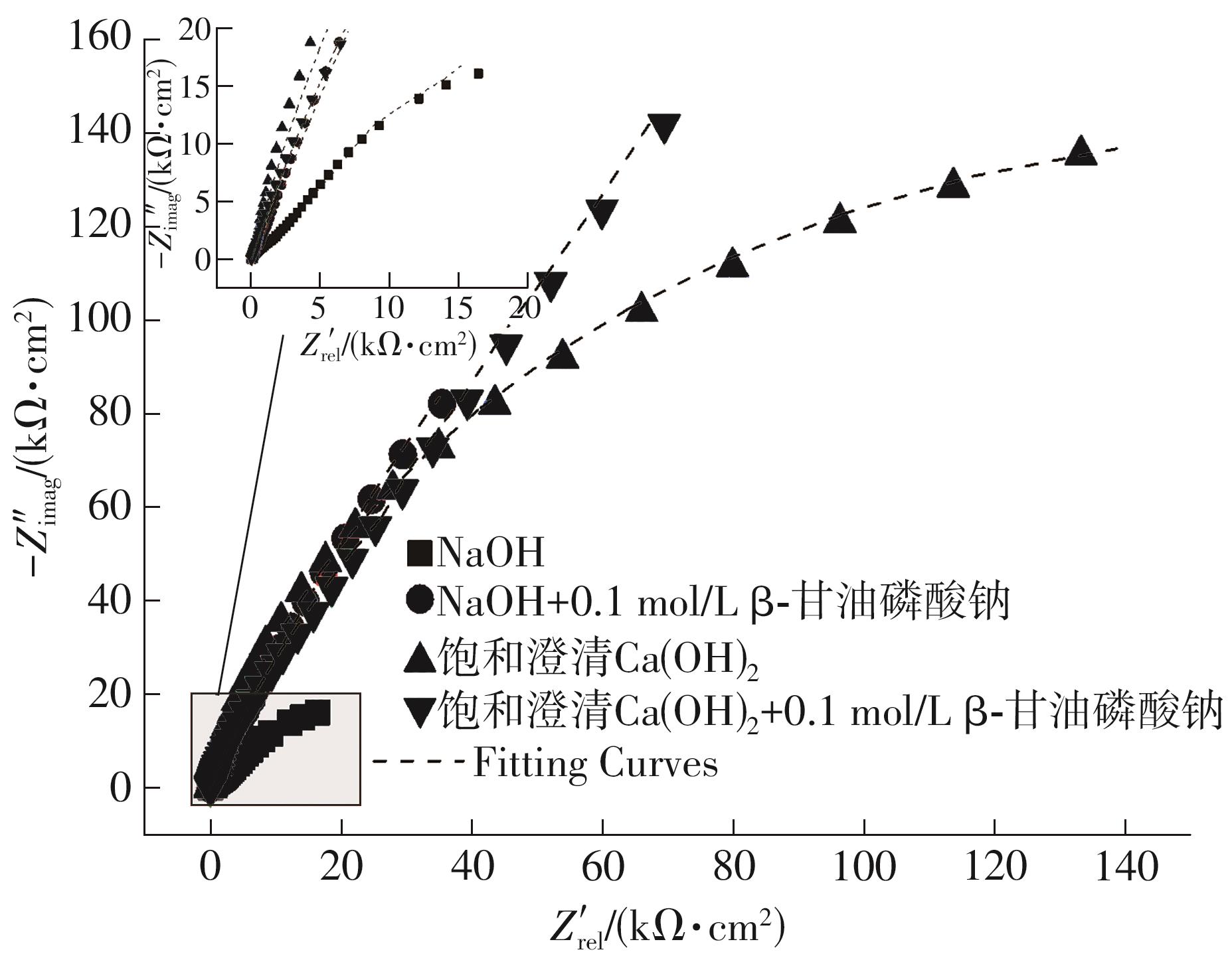
EIS parameters and inhibition efficiency of steel bars in different solutions under the 0.02 mol/L NaCl condition"
| 样品 | 电化学参数 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Rs/Ω | Rf / (kΩ·cm2) | CPEf / (μF·cm-2) | nf | Rct / (kΩ·cm2) | CPEdl / (μF·cm-2) | ndl | W | χ2/10-3 | ηE1/% | ηE2/% | |
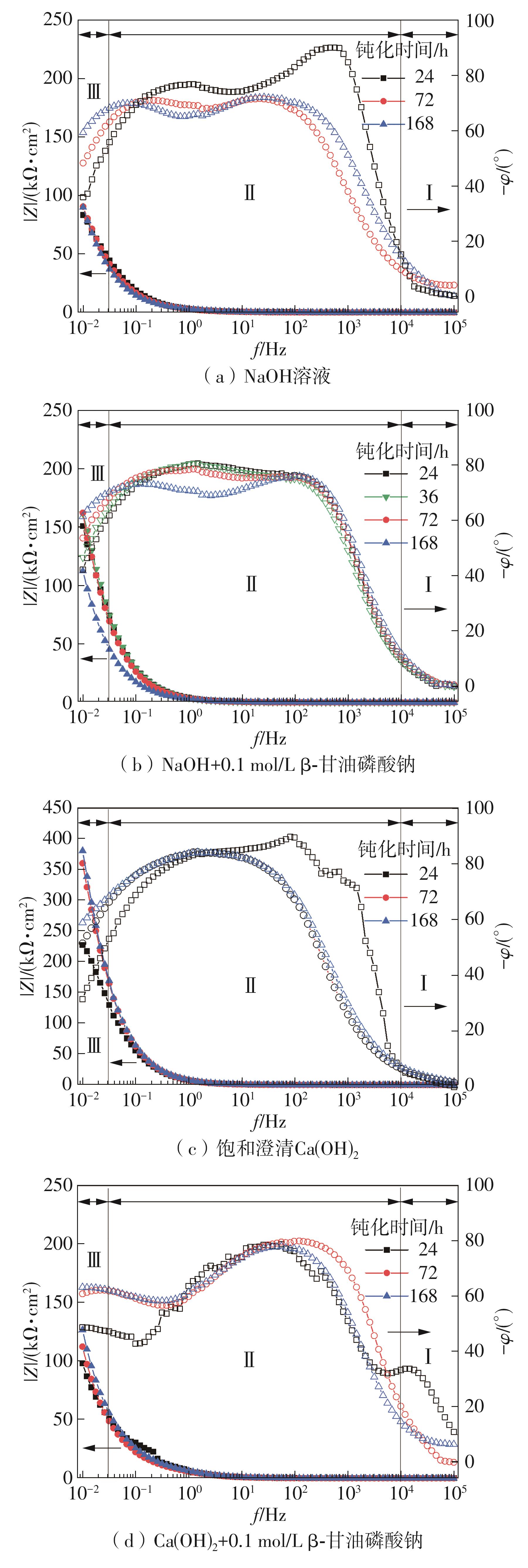
| NaOH溶液 | 25.18 | — | — | — | 2.04 | 74.873 | 0.81 | 0.005 5 | 0.903 | — | — |
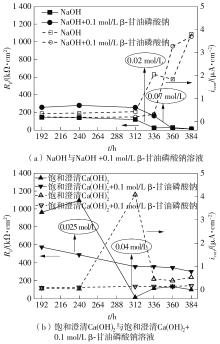
| NaOH+0.1 mol/L β-甘油磷酸钠 | 16.55 | 2.09 | 58.252 | 0.84 | 999.55 | 42.002 | 0.77 | — | 0.574 | 99.80 | — |
| 饱和澄清Ca(OH)2 | 41.49 | — | — | — | 345.93 | 40.558 | 0.87 | — | 5.669 | — | 99.41 |
| 饱和澄清Ca(OH)2+0.1 mol/L β-甘油磷酸钠 | 14.54 | — | — | — | 354.87 | 46.955 | 0.83 | 0.004 2 | 13.123 | 2.52 | — |
| 1 | PRADHAN B .A study on effectiveness of inorganic and organic corrosion inhibitors on rebar corrosion in concrete:a review[J].Materials Today:Proceedings,2022,65(2):1360-1366. |
| 2 | XU P, JIANG L, GUO M Z,et al .Influence of sulfate salt type on passive film of steel in simulated concrete pore solution[J].Construction and Building Materials,2019,223:352-359. |
| 3 | SABEL C F, VICTOR D G .Governing global problems under uncertainty:making bottom-up climate policy work[J].Climatic Change,2015,144(1):15-27. |
| 4 | KAMARUZZAMAN W M I W M, NASIR N A M, HAMIDI N A S M,et al .Frontiers in organic corrosion inhibitors for chloride and acidic media:a review[J].Journal of Bio- and Tribo-Corrosion,2022,8(2):1-18. |
| 5 | ANGST U M .Challenges and opportunities in corrosion of steel in concrete[J].Materials and Structures,2018,51(1):1-20. |
| 6 | BOLZONI F, BRENNA A, ORMELLESE M .Recent advances in the use of inhibitors to prevent chloride-induced corrosion in reinforced concrete[J].Cement and Concrete Research,2022,154:1-9. |
| 7 | RAJA P B, GHOREISHIAMIRI S, ISMAIL M .Natural corrosion inhibitors for steel reinforcement in concrete-a review[J].Surface Review and Letters,2015,22(3):1-8. |
| 8 | BOLZONI F, COPPOLA L, GOIDANICH S,et al .Corrosion inhibitors in reinforced concrete structures Part 1:preventative technique [J].Corrosion Engineering,Science and Technology,2013,39(3):219-228. |
| 9 | VERMA C, OLASUNKANMI L O, EBENSO E E,et al .Substituents effect on corrosion inhibition performance of organic compounds in aggressive ionic solutions:a review[J].Journal of Molecular Liquids,2018,251:100-118. |
| 10 | GENG J, LIU J, YAN J,et al .Chemical composition of corrosion products of rebar caused by carbonation and chloride[J].International Journal of Corrosion,2018,2018:1-7. |
| 11 | XU Y, GUO X, CHEN N,et al .Synthesis and corrosion inhibitory effect of a novel β-amino alcohol compound[J].Colloids and Surfaces A Physicochemical and Engineering Aspects,2021,610(3):1-9. |
| 12 | LOTHENBACH B, WINNEFELD F, ALDER C,et al .Effect of temperature on the pore solution,microstructure and hydration products of Portland cement pastes[J].Cement and Concrete Research,2007,37(4):483-491. |
| 13 | GHODS P, ISGOR O B, CARPENTER G J C,et al .Nano-scale study of passive films and chloride-induced depassivation of carbon steel rebar in simulated concrete pore solutions using FIB/TEM[J].Cement and Concrete Research,2013,47:55-68. |
| 14 | 赵冰,杜荣归,林昌健 .三种有机缓蚀剂对钢筋阻锈作用的电化学研究[J].电化学,2005,11(4):382-386. |
| ZHAO Bing, DU Ronggui, LIN Changjian .A study of three corrosion inhibitors for reinforcing steel in SPS solution by electrochemical methods[J].Electrochemistry,2005,11(4):382-386. | |
| 15 | NER B E .Corrosion inhibitors for reinforced concrete-an EFC state of the art report[J].Corrosion of Reinforcement in Concrete:Monitoring,Prevention and Rehabilitation Techniques,2014,38:170-184. |
| 16 | STEFANONI M, ANGST U, ELSENER B .Influence of calcium nitrate and sodium hydroxide on carbonation-induced steel corrosion in concrete[J].Corrosion,2019,75(7):737-744. |
| 17 | SINGH J K, YANG H M, LEE H S,et al .Role of L-arginine on the formation and breakdown of passive film onto the steel rebars surface in chloride contaminated concrete pore solution[J].Journal of Molecular Liquids,2021,337:1-15. |
| 18 | TOUJAS S, VAZQUEZ M, VALCARCE M B .Unexpected effect of citrate ions on the corrosion process of carbon steel in alkaline solutions[J].Corrosion Science,2017,128(11):94-99. |
| 19 | 周霄骋,穆松,马麒,等 .混凝土模拟孔溶液中有机钢筋阻锈剂的加速评价及等效性分析[J].硅酸盐学报,2021,49(8):1713-1721. |
| ZHOU Xiaocheng, MU Song, MA Qi,et al .Accelerated evaluation of organic steel corrosion inhibitor in simulated pore solution of concrete and its equivalence analysis[J].Journal of the Chinese Ceramic Society,2021,49(8):1713-1721. | |
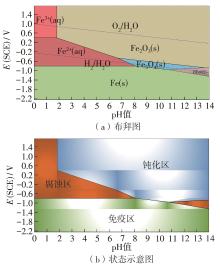
| 20 | POURBAIX M .Applications of electrochemistry in corrosion science and in practice[J].Corrosion Science,1974,14(1):25-82. |
| 21 | PEDEFERRI P, PEDEFERRI P .Pourbaix diagrams[M].Corrosion Science and Engineering,2018:57-72. |
| 22 | THOMPSON W T, KAYE M H, BALE C W,et al .Pourbaix diagrams for multielement systems[J].Uhlig’s Corrosion Handbook,2011,3:103-109. |
| 23 | BEVERSKOG B, PUIGDOMENECH I .Revised pourbaix diagrams for iron at 25-300 C[J].Corrosion Science,1996,38(12):2121-2135. |
| 24 | MCCAFFERTY E .Introduction to corrosion science[M].New York,NY:Springer,2010. |
| 25 | DAS J K, PRADHAN B .Effect of cation type of chloride salts on corrosion behaviour of steel in concrete powder electrolyte solution in the presence of corrosion inhibitors[J].Construction and Building Materials,2019,208(30):175-191. |
| 26 | ZHENG H, DAI J G, POON C S,et al .Influence of calcium ion in concrete pore solution on the passivation of galvanized steel bars[J].Cement and Concrete Research,2018,108:46-58. |
| 27 | WANG D, MING J, SHI J .Enhanced corrosion resistance of rebar in carbonated concrete pore solutions by Na2HPO4 and benzotriazole[J].Corrosion Science,2020,174:1-12. |
| 28 | TEYMOURI F, SAMIEIi I, ALLAHKARAM S R,et al .Passive film alteration of reinforcing steel through [MoO42-]/[RCOO-]interfacial co-interaction for enhanced corrosion resistance in chloride contaminated concrete pore solution[J].Journal of Molecular Liquids,2022,356:1-18. |
| 29 | ZHAO Y, PAN T, YU X,et al .Corrosion inhibition efficiency of triethanolammonium dodecylbenzene sulfonate on Q235 carbon steel in simulated concrete pore solution[J].Corrosion Science,2019,158:1-12. |
| 30 | SHI J, SUN W, JIANG J,et al .Influence of chloride concentration and pre-passivation on the pitting corrosion resistance of low-alloy reinforcing steel in simulated concrete pore solution[J].Construction & Building Materials,2016,111(15):805-813. |
| 31 | QIANG Y, ZHANG S, XU S,et al .The effect of 5-nitroindazole as an inhibitor for the corrosion of copper in a 3.0% NaCl solution[J].RSC Adv,2015,5(78):63866-63873. |
| 32 | SáNCHEZ M, GREGORI J, ALONSO M C,et al .Anodic growth of passive layers on steel rebars in an alkaline medium simulating the concrete pores[J].Electrochimica Acta,2006,52(1):47-53. |
| 33 | XU W, DAUB K, ZHANG X,et al .Oxide formation and conversion on carbon steel in mildly basic solutions[J].Electrochimica Acta,2009,54(24):5727-5738. |
| 34 | FREIRE L, NóVOA X R, MONTEMOR M F,et al .Study of passive films formed on mild steel in alkaline media by the application of anodic potentials[J].Materials Chemistry and Physics,2009,114(2/3):962-972. |
| 35 | WANG F, ZHANG Z, WU S,et al .Effect of inhibitor on adsorption behavior and mechanism of micro-zone corrosion on carbon steel[J].Materials,2019,12(12):1901-1911. |
| [1] | SHEN Lin CHEN Dingshi LI Wanheng ZHAO Shangchuan. Mesoscopic and Stochastic Research on Chloride Ion Transport in Unsaturated Concrete [J]. Journal of South China University of Technology (Natural Science Edition), 2018, 46(11): 76-82. |
| [2] | Xie Yah Wu Xiao-mei Fan Yue-ming Yu Qi-jun Chen Ying-yun. Influence of Doped Chloride Ions on Steel Corrosion and Solidification of Chloride Ions by Different Materials [J]. Journal of South China University of Technology (Natural Science Edition), 2009, 37(8): 132-139. |
| [3] | Ren Bi-ye Shi Lu Cheng Zhi-yu Shan Hua-zhong Li Chong-qing Tong Zhen. Synthesis and Electrochemical Behaviour of Dendritic Amphiphilic Molecules with Ferroncenyl Substituents [J]. Journal of South China University of Technology (Natural Science Edition), 2009, 37(12): 28-31,36. |
| [4] | Lin Bi-lan Lu Jin-tang Kong Gang. Electrochemical Behaviors of PhosphatedlMolybdate Post-Sealed Hot-Dipped Galvanized Steel Sheet [J]. Journal of South China University of Technology (Natural Science Edition), 2007, 35(8): 93-97,108. |
| [5] | Liang Song Yang Yi-bo Mo Hai-hong Chen You-wen. Field Experiments of Anti-Seawater Corrosion Concrete with a Lot of Slag in Tidal Region [J]. Journal of South China University of Technology (Natural Science Edition), 2005, 33(7): 88-91. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||