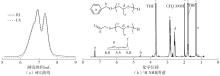
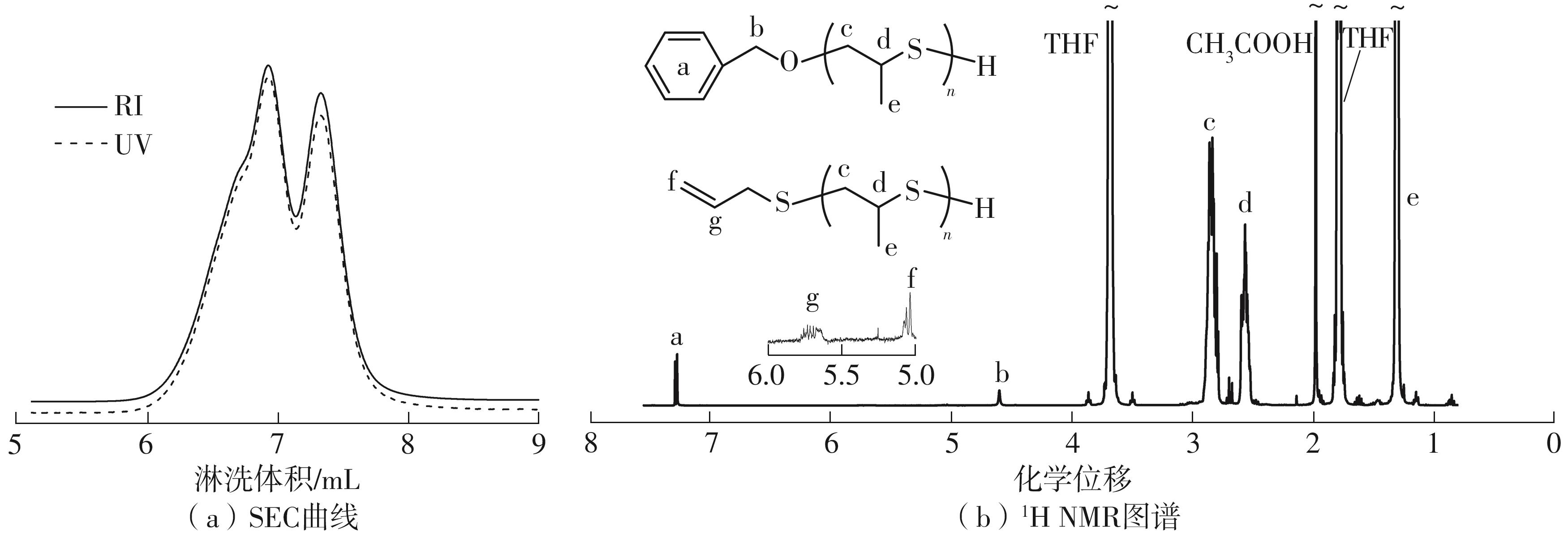
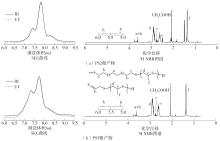
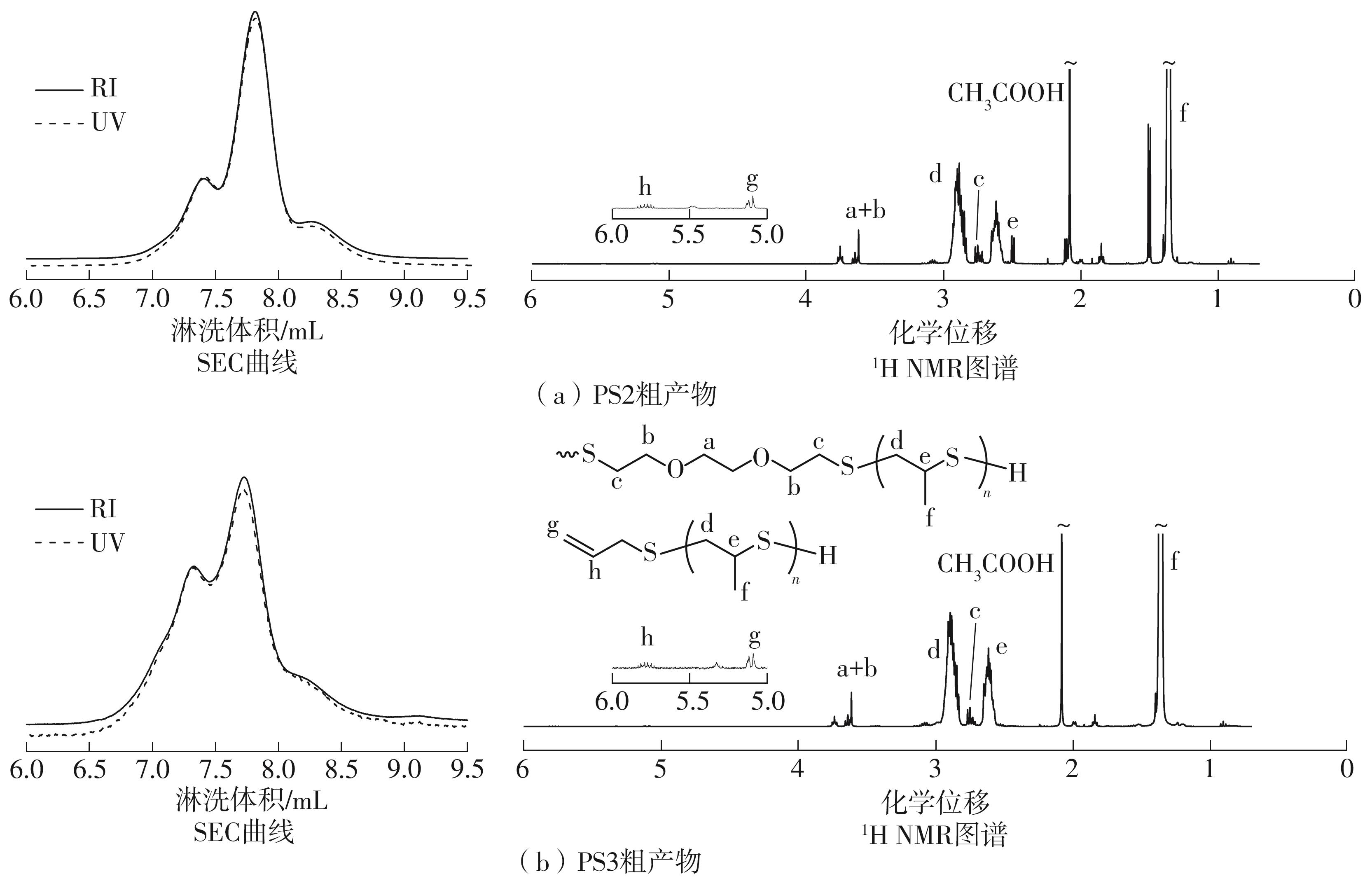
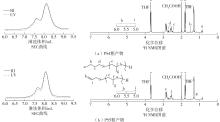
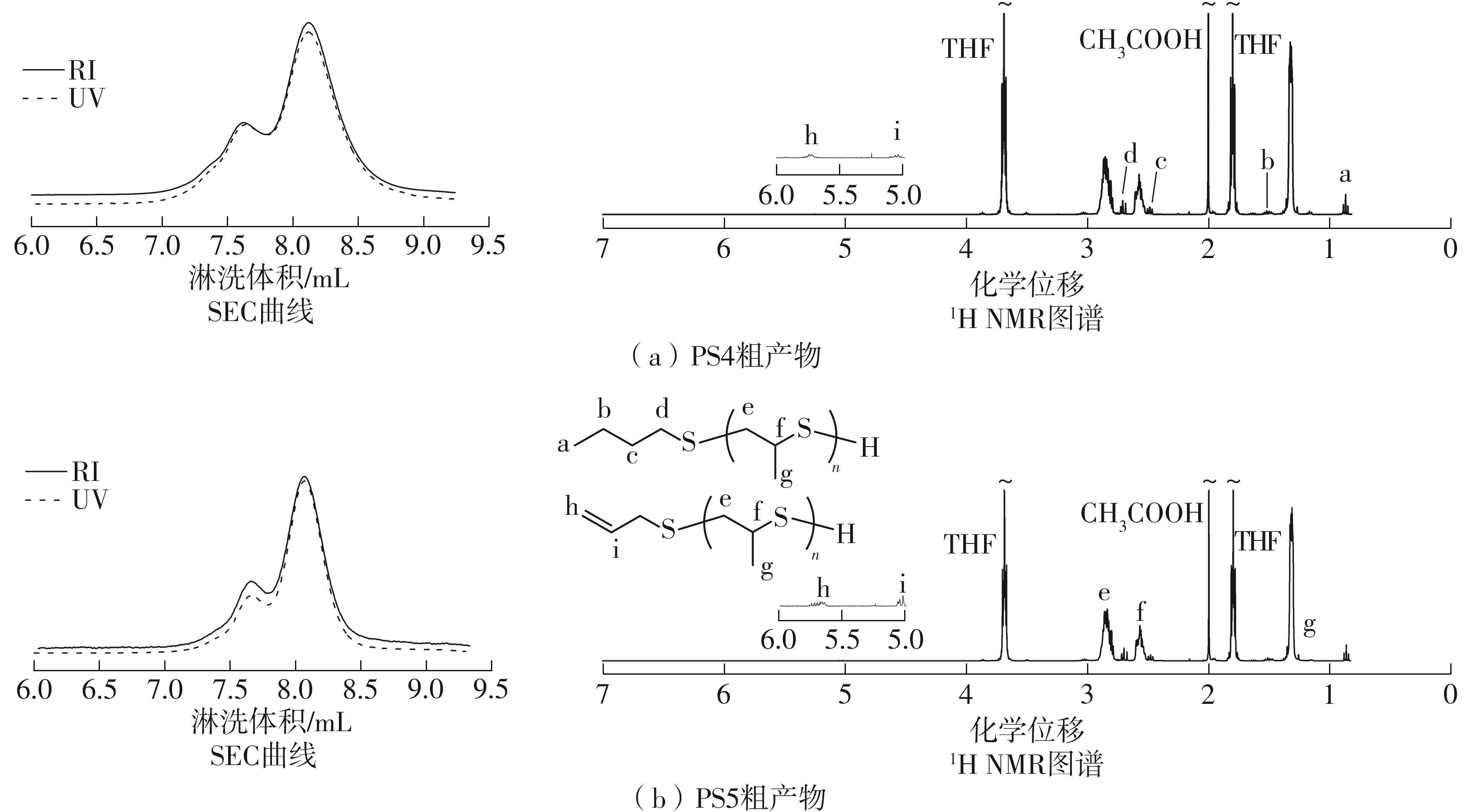
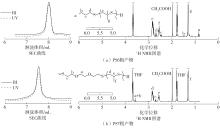
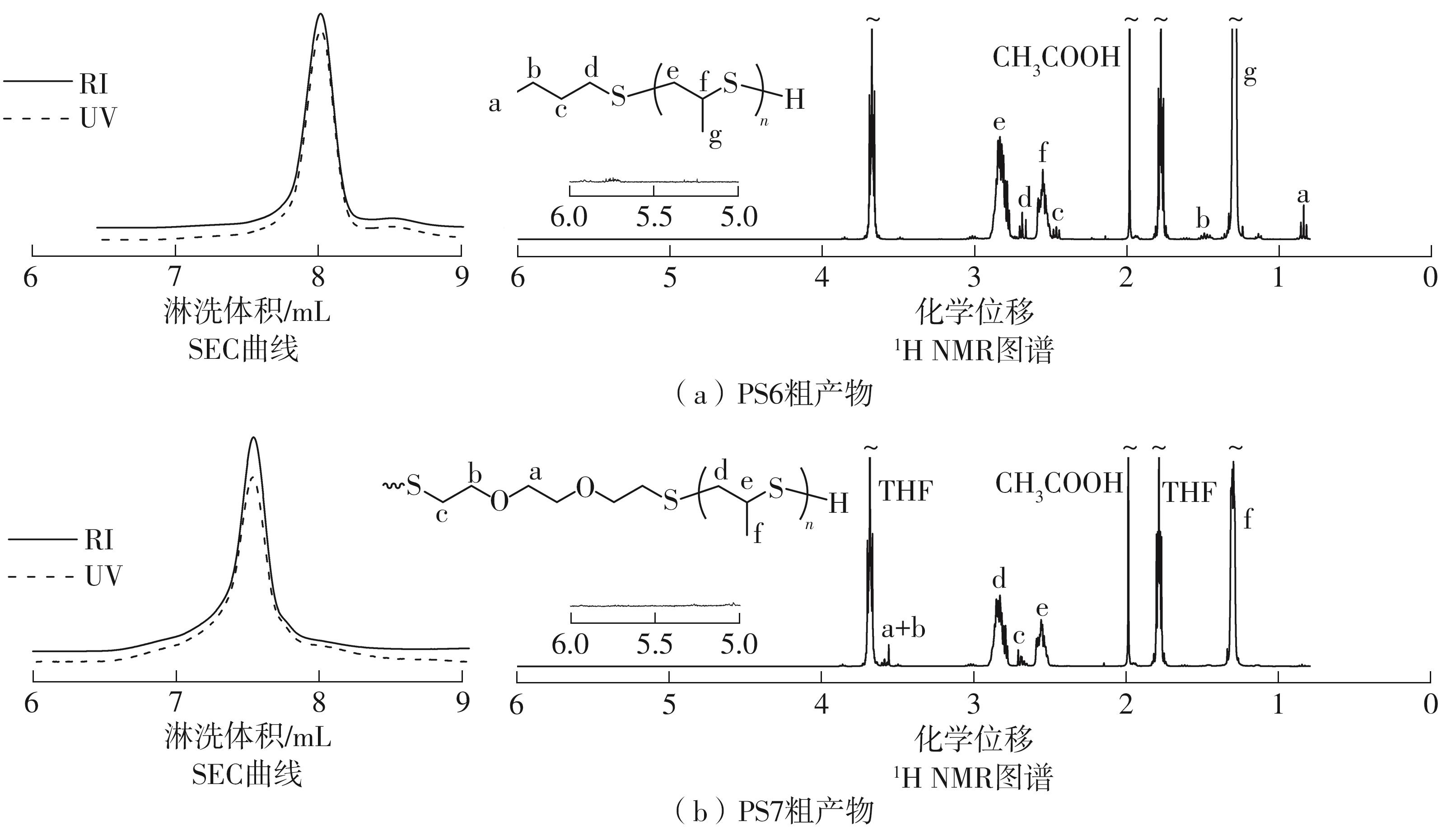
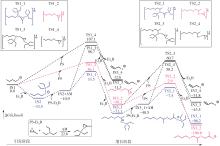
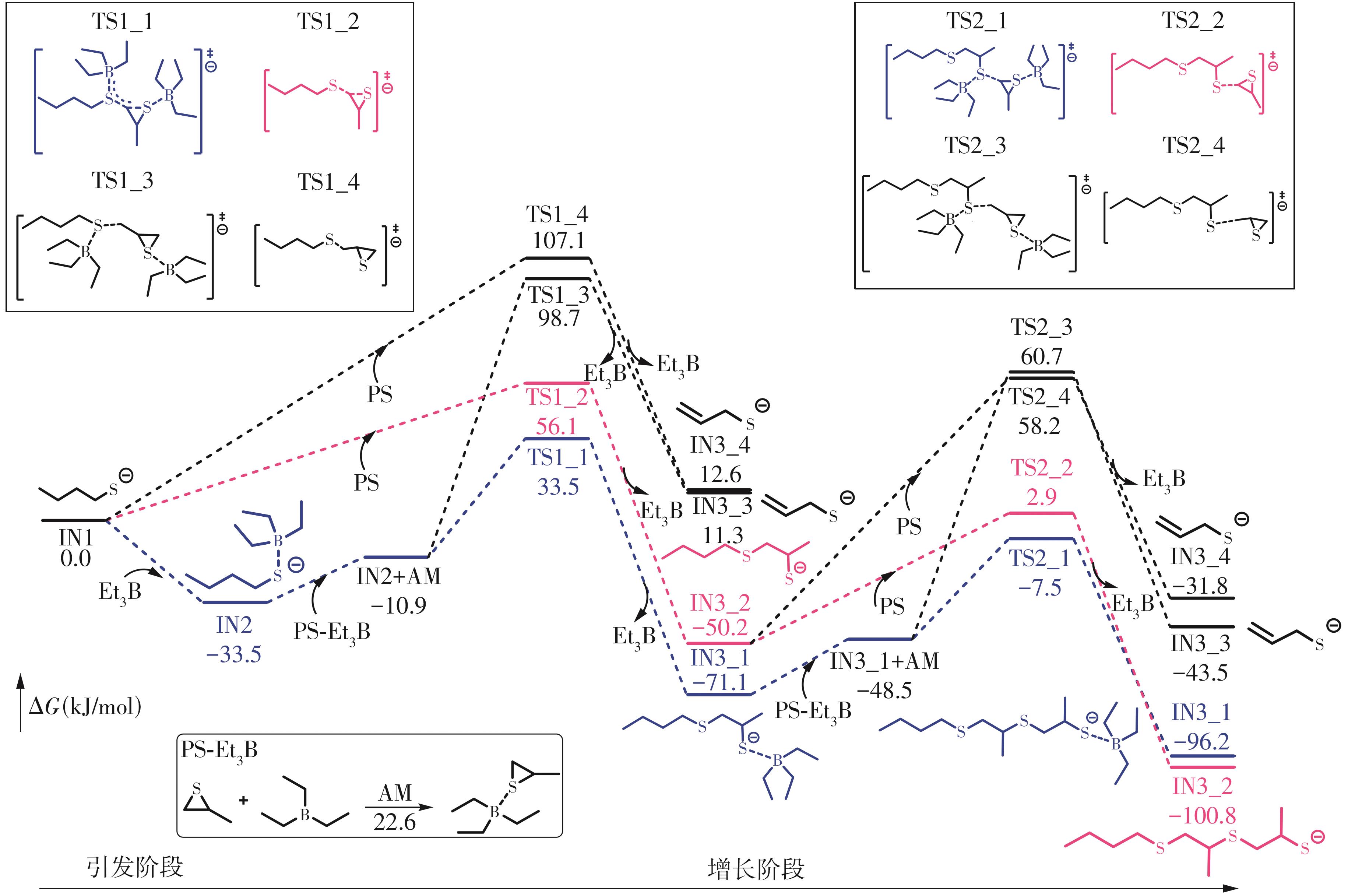
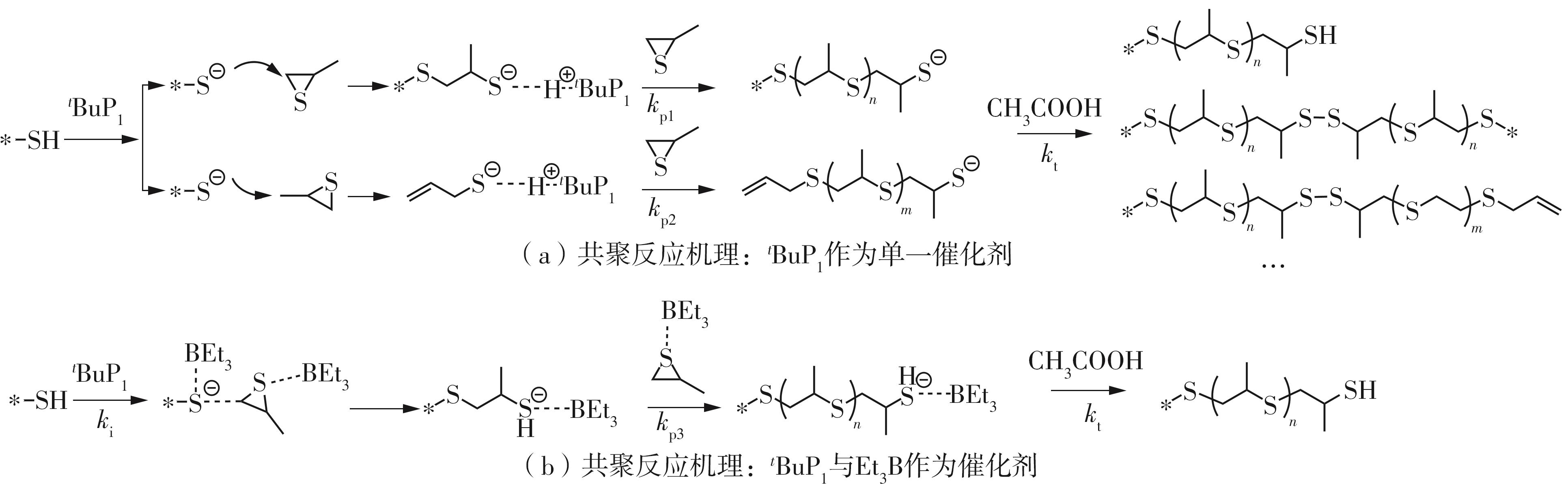
| [1] |
PENCZEK S, CYPRYK M, PRETULA J,et al .Elemental sulfur and cyclic sulfides. Homo- and copolymerizations.Kinetics,thermodynamics and DFT analysis[J].Progress in Polymer Science,2024,152:101818/1-32.
|
| [2] |
MUTLU H, CEPER E B, LI X,et al .Sulfur chemistry in polymer and materials science[J].Macromolecular Rapid Communications,2019,40(1):1800650/1-51.
|
| [3] |
YUE T, REN W, LU X .Copolymerization involving sulfur-containing monomers[J].Chemical Reviews,2023,123(24):14038-14083.
|
| [4] |
IIMORI H, SHIBASAKI Y, ANDO S,et al .Nonstoichiometric polycondensation I. Synthesis of polythioether from dibromomethane and 4,4′‐thiobisbenzenethiol[J].Macromolecular Symposia,2003,199(1):23-36.
|
| [5] |
CLANCY S, SHRIVER D F, OCHRYMOWYCZ L A .Preparation and characterization of polymeric solid electrolytes from poly(alkylene sulfides) and silver salts[J].Macromolecules,1986,19(3):606-611.
|
| [6] |
DAGLAR O, ÇAKMAKÇI E, HIZAL G,et al .Extremely fast synthesis of polythioether based phase change materials (PCMs) for thermal energy storage[J].European Polymer Journal,2020,130:109681/1-11.
|
| [7] |
ELEMM E, SENSFUß S .Untersuchungen zum selbstinitiierungs‐mechanismus der thiol/En‐polymerisation[J].Die Makromolekulare Chemie,1991,192(1):159-164.
|
| [8] |
EVANS R A, RIZZARDO E .Free-radical ring-opening polymerization of cyclic allylic sulfides[J].Macromole-cules,1996,29(22):6983-6989.
|
| [9] |
GAO T, XIA X, TAJIMA K,et al .Polyether/polythioether synthesis via ring-opening polymerization of epoxides and episulfides catalyzed by alkali metal carboxylates[J].Macromolecules,2022,55(21):9373-9383.
|
| [10] |
LIU Y, CHEN Y, ZHANG Y,et al .Density-adjustable bio-based polysulfide composite prepared by inverse vulcanization and bio-based fillers[J].Polymers,2020,12(9):2127/1-15.
|
| [11] |
GE C, ZHU J, WU G,et al .ROS-responsive selenopolypeptide micelles:preparation,characterization,and controlled drug release[J].Biomacromolecules,2022,23(6):2647-2654.
|
| [12] |
YANG W, ZHOU W, DING N,et al .Biobased photothermal responsive shape memory polythioether/MXene nanocomposites with self-extinguishing performance[J].Chemical Engineering Journal,2024,497:154591/1-13.
|
| [13] |
WANG C, LIANG J, ZHAO Y,et al .All-solid-state lithium batteries enabled by sulfide electrolytes:from fundamental research to practical engineering design[J].Energy & Environmental Science,2021,14(5):2577-2619.
|
| [14] |
KAMBER N E, JEONE W, WAYMOUTH R M,et al .Organocatalytic ring-opening polymerization[J].Chemical Reviews,2007,107(12):5813-5840.
|
| [15] |
SONG Q, PASCOUAU C, ZHAO J,et al .Ring-opening polymerization of γ-lactones and copolymerization with other cyclic monomers[J].Progress in Polymer Science,2020,110:101309/1-27.
|
| [16] |
KIESEWETTER M K, SHIN E J, HEDRICK J L,et al .Organocatalysis: opportunities and challenges for polymer synthesis[J].Macromolecules,2010,43(5):2093-2107.
|
| [17] |
ZHANG C, ZHANG X .Chemoselective coupling of CS2 and epoxides for producing poly(thioether)s and COS via oxygen/sulfur atom exchange[J].Macromole-cules,2020,53(1):233-239.
|
| [18] |
YANG H, HUANG J, SONG Y,et al .Anionic hybrid copolymerization of sulfur with acrylate: strategy for synthesis of high-performance sulfur-based polymers[J].Journal of the American Chemical Society,2023,145(26):14539-14547.
|
| [19] |
CHEN Y, LIU S, ZHAO J,et al .Chemoselective polymerization of epoxides from carboxylic acids: direct access to esterified polyethers and biodegradable polyurethanes[J].ACS Macro Letters,2019,8(12):1582-1587.
|
| [20] |
XIA Y, SCHEUTZ G M, EASTERLING C P,et al .Hybrid block copolymer synthesis by merging photoiniferter and organocatalytic ring‐opening polymerizations[J].Angewandte Chemie International Edition,2021,60(34):18537-18541.
|
| [21] |
HONG X, LIU S, PANG J,et al .Polyglycidamides:from backbone‐promoted amidation to degradable polyether with wide‐range LCST[J].Angewandte Chemie International Edition,2025,64(7):e202419978/1-10.
|
| [22] |
YANG J, WU H, LI Y,et al .Perfectly alternating and regioselective copolymerization of carbonyl sulfide and epoxides by metal‐free Lewis pairs[J].Angewandte Chemie International Edition,2017,56(21):5774-5779.
|
| [23] |
LUYER S L, GUÉGAN P, ILLY N .Episulfide anionic ring-opening polymerization initiated by alcohols and primary amines in the presence of γ-thiolactones[J].Macromolecules,2022,55(13):5430-5440.
|
| [24] |
CHEN Y, SHEN J, LIU S,et al .High efficiency organic Lewis pair catalyst for ring-opening polymerization of epoxides with chemoselectivity[J].Macromolecules,2018,51(20):8286-8297.
|
| [25] |
ZHANG D, FENG X, GNANOU Y,et al .Theoretical mechanistic investigation into metal-free alternating copolymerization of CO2 and epoxides: the key role of triethylborane[J].Macromolecules,2018,51(15):5600-5607.
|
| [26] |
LIU L, ZHAO J, ZHANG G .Chemoselectivity streamlines the approach to linear and Y-shaped thiol-polyethers starting from thiocarboxylic acids[J].ACS Macro Letters,2023,12(9):1185-1192.
|
| [27] |
NAKANO K, TATSUMI G, NOZAKI K .Synthesis of sulfur-rich polymers: copolymerization of episulfide with carbon disulfide by using[PPN]Cl/(salph)Cr(Ⅲ)Cl System[J].Journal of the American Chemical Society,2007,129(49):15116-15117.
|
| [28] |
ZHANG C, YANG Y, HE J .Direct transformation of living anionic polymerization into RAFT-based polymerization[J].Macromolecules,2013,46(10):3985-3994.
|