华南理工大学学报(自然科学版) ›› 2026, Vol. 54 ›› Issue (2): 102-111.doi: 10.12141/j.issn.1000-565X.250302
鸡肉肽-亚铁螯合物对缺铁性贫血小鼠的影响
刘蕊莉1, 宋雪盈1, 苗晋鑫2, 赵谋明1, 苏国万1
- 1.华南理工大学 食品科学与工程学院,广东 广州 510640
2.河南中医药大学 中医药科学院,河南 郑州 450046
Effect of Chicken Peptide-Ferrous Chelate on Iron Deficiency Anemia in Mice
LIU Ruili1, SONG Xueying1, MIAO Jinxin2, ZHAO Mouming1, SU Guowan1
- 1.School of Food Science and Engineering,South China University of Technology,Guangzhou 510640,Guangdong,China
2.Academy of Chinese Medical Sciences,Henan University of Chinese Medicine,Zhengzhou 450046,Henan,China
摘要:
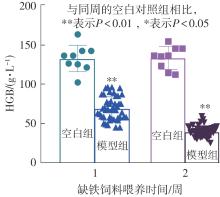
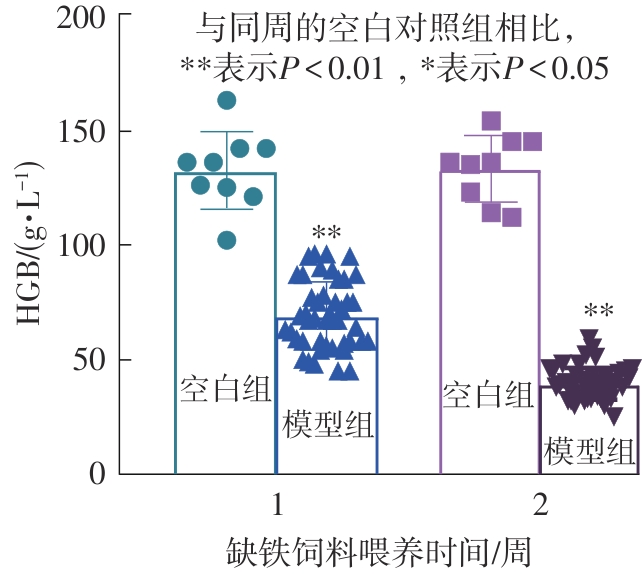
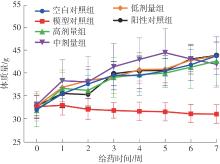
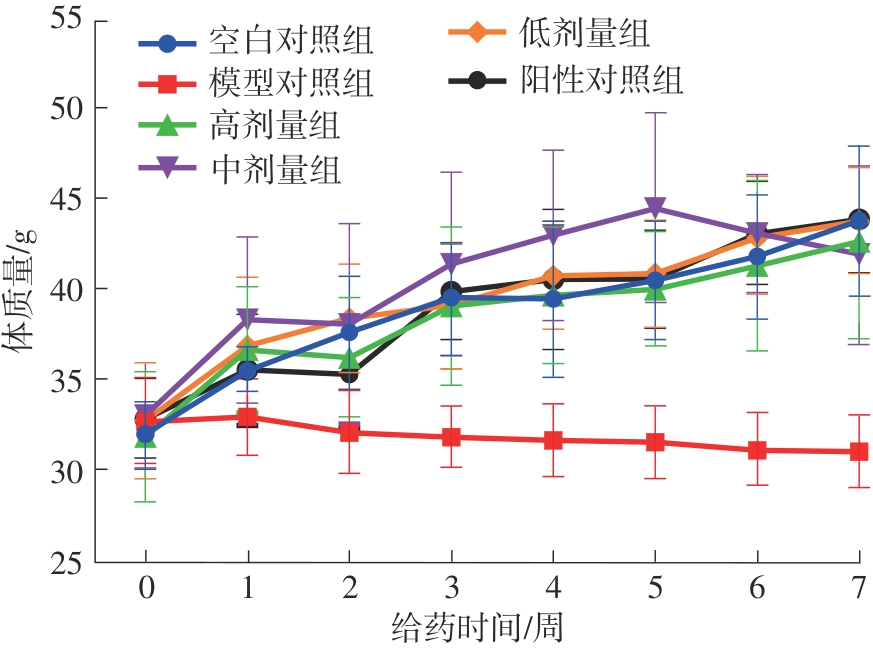
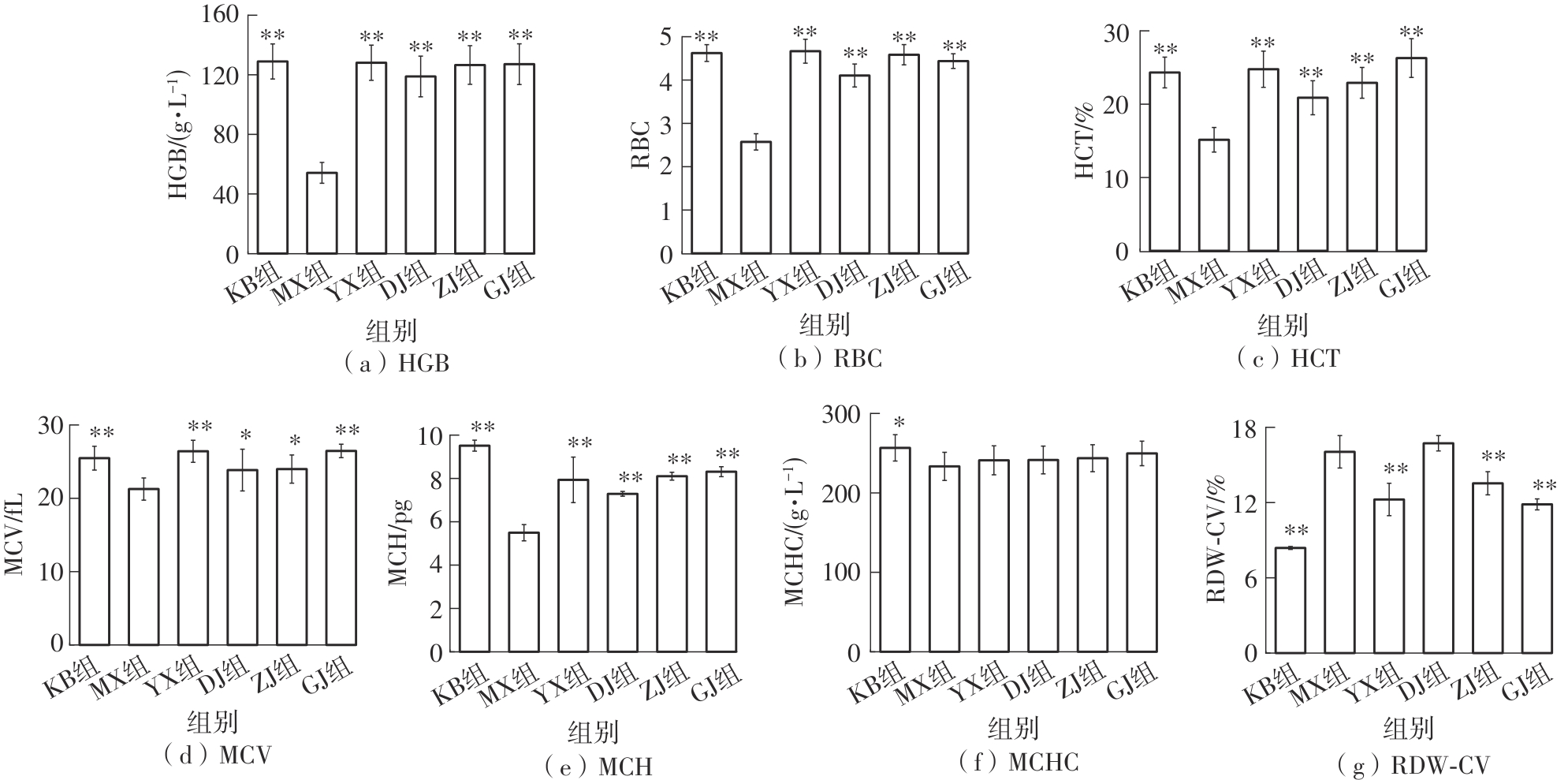
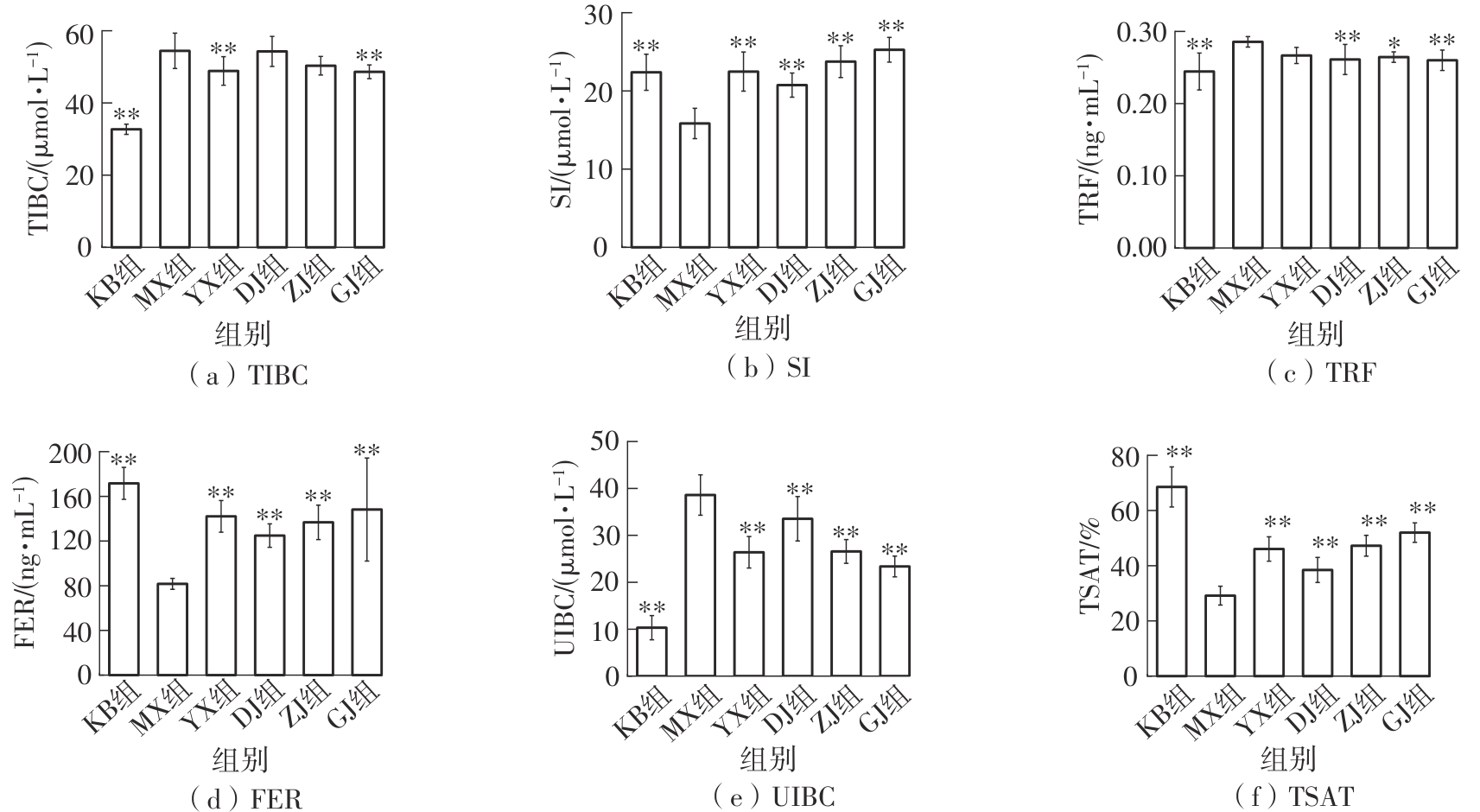
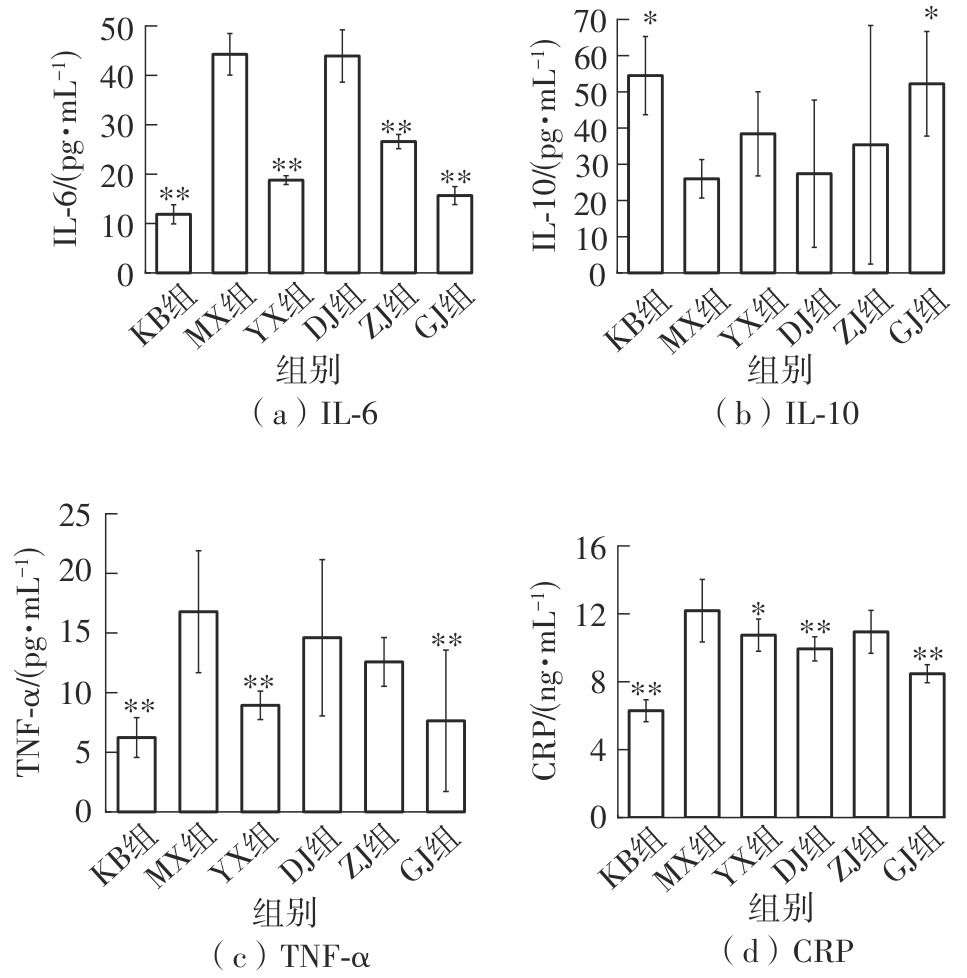
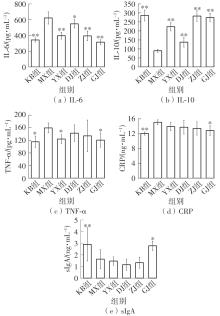
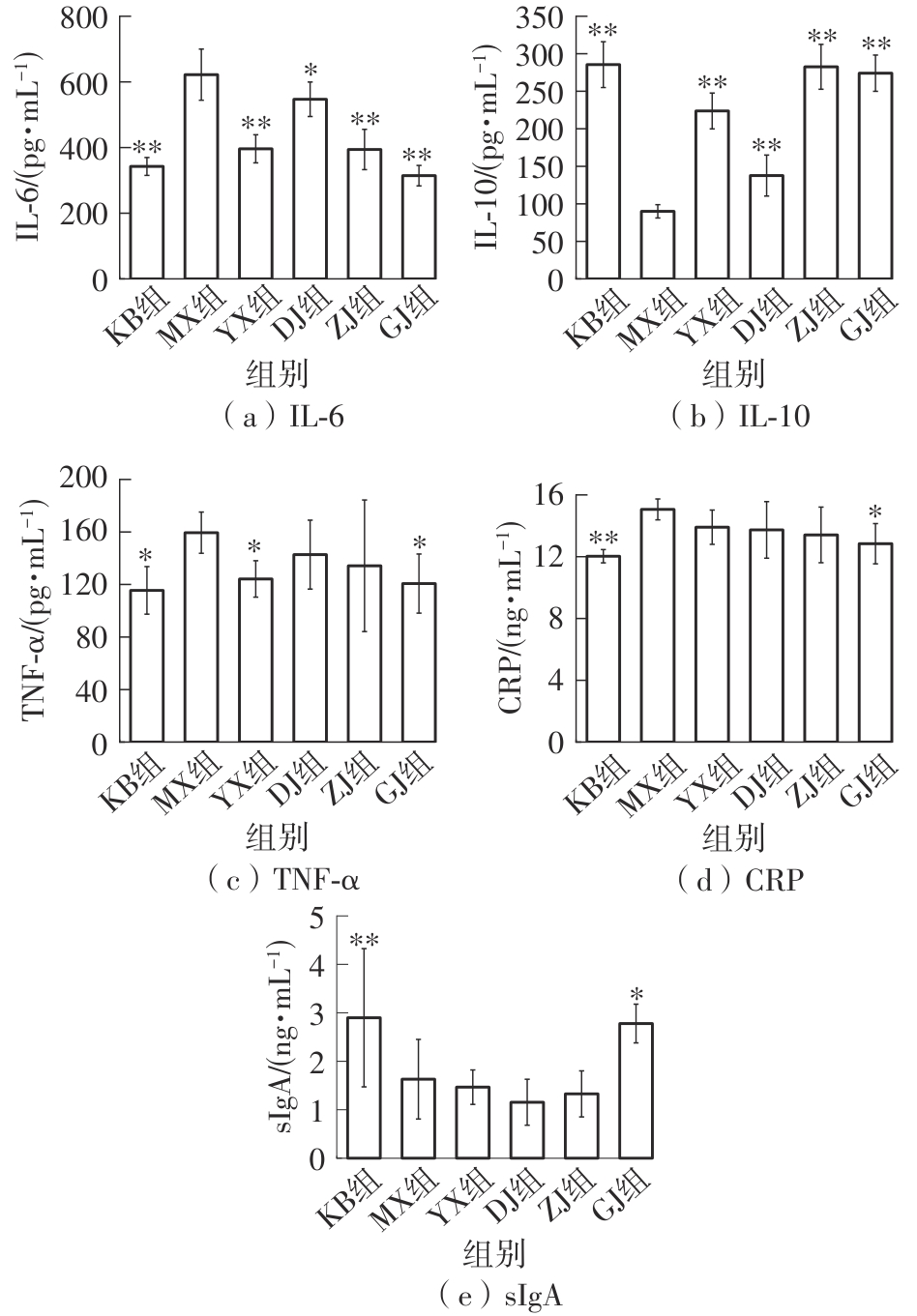
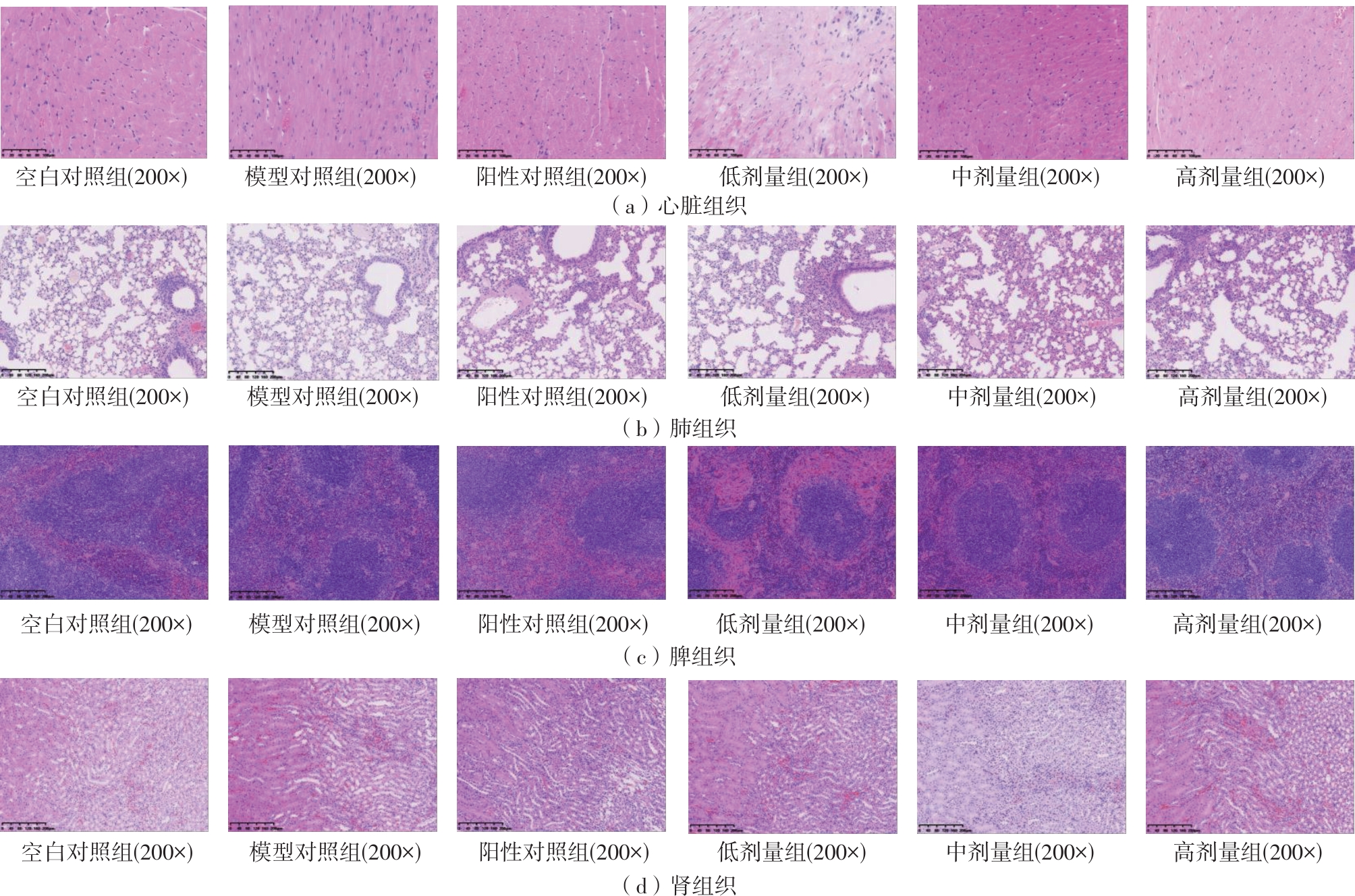
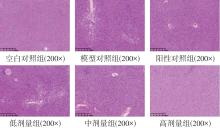
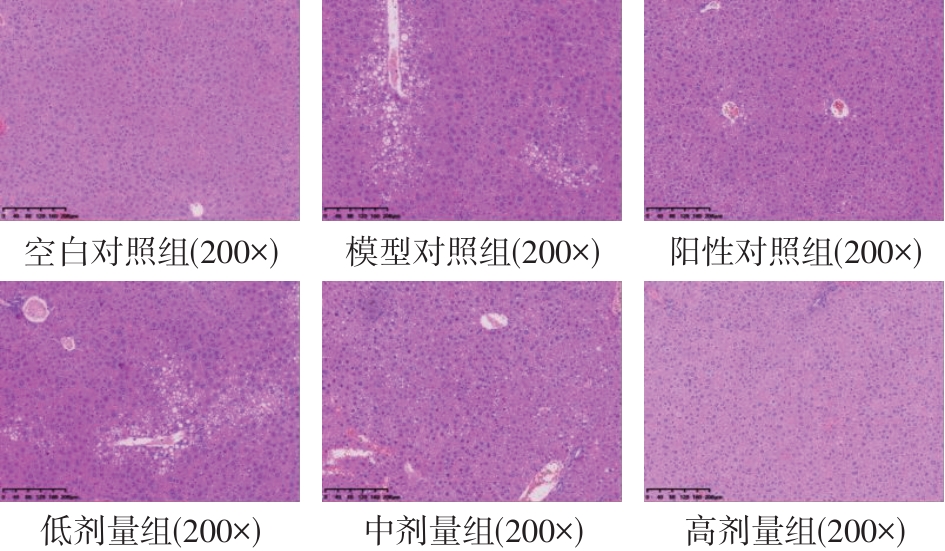
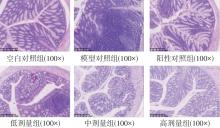
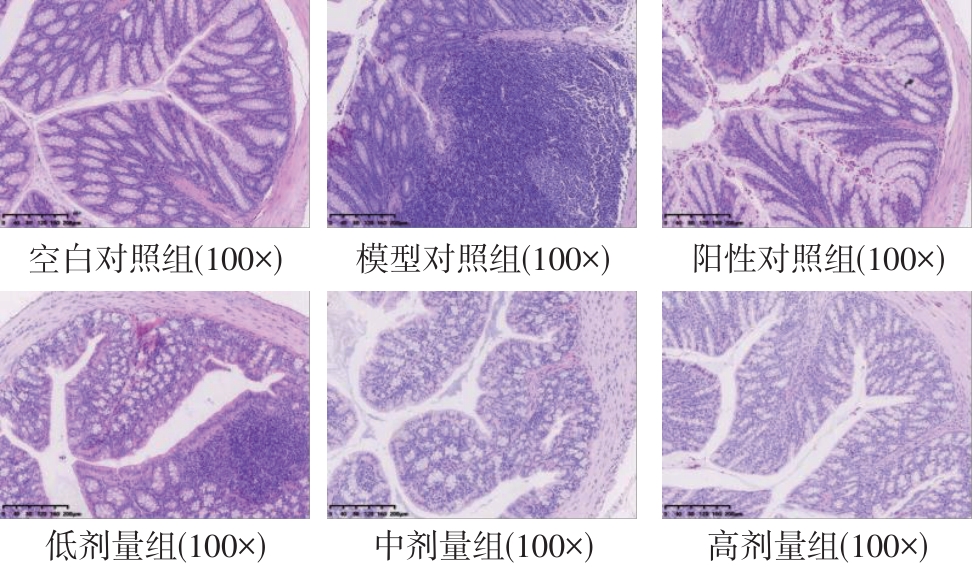
该文系统探讨了鸡肉肽-亚铁螯合物(CMP-Fe)对缺铁性贫血(IDA)小鼠贫血症状的改善作用,并从体质量变化、血常规参数、铁代谢指标、炎症反应及组织病理学等多个维度,综合评价其干预效应。实验结果表明:与模型组相比,CMP-Fe各剂量组(按小鼠体质量计,小、中、高剂量组分别喂食Fe 1.0、2.0、3.0 mg/kg)的小鼠体质量、血常规指标(包括红细胞计数RBC、血红蛋白HGB、红细胞压积HCT、平均红细胞体积MCV、平均红细胞血红蛋白含量MCH、平均红细胞血红蛋白浓度MCHC、红细胞分布宽度-变异系数RDW-CV)及血清铁代谢指标(包括血清铁SI、总铁结合力TIBC、转铁蛋白TRF、铁蛋白FER、转铁蛋白饱和度TSAT、不饱和铁结合力UIBC)均得到显著改善,且高剂量组效果最为突出。CMP-Fe对RBC、HGB、HCT等红细胞系指标的改善作用与阳性对照组相当,并呈现明显的剂量依赖性(小、中、高剂量梯度)。在炎症调控方面,CMP-Fe可抑制血清及结肠促炎因子(IL-6、TNF-α和CRP)的产生,同时提升抑炎因子IL-10及肠道黏膜免疫标志物sIgA的水平。具体而言,CMP-Fe组小鼠结肠组织中IL-6、TNF-α及CRP等促炎因子水平显著低于模型对照组(P < 0.05),提示其可有效调节IDA伴随的炎症反应;尤其在高剂量CMP-Fe干预下,小鼠sIgA水平的恢复效果优于模型对照组(P< 0.05),甚至超过阳性对照组。组织病理学检查显示,CMP-Fe对小鼠的心、肺、脾、肾等器官无明显病理性损伤,表明其具有良好的生物安全性;同时,该螯合物还能显著缓解铁缺乏引起的肠道及肝脏组织病理损伤。综上所述,CMP-Fe可有效改善IDA小鼠的铁代谢紊乱、抑制炎症反应、减轻肠道及肝脏组织损伤,且安全性良好,是一种具有开发潜力的新型有机补铁剂,可用于猫、狗等多种宠物专用粮及营养补充剂的产品开发。
中图分类号: