

Journal of South China University of Technology(Natural Science) >
Controlled Anionic Polymerization of Propylene Sulfide and Strategy for Synthesis of Sulfur-Rich Copolymers
Received date: 2025-04-21
Online published: 2025-05-07
Supported by
the National Natural Science Foundation of China(52263001);the Natural Science Foundation of Qinghai Province(2022-ZJ-944Q)
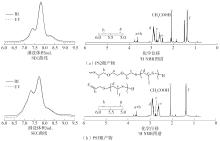
Ring-opening polymerization of cyclic sulfur compounds is one of the important methods to synthesize sulfur-containing polymers. However, in traditional anionic polymerization techniques, the high reactivity of sulfur-centered anions tends to induce chain transfer side reactions, leading to products with broad molecular mass distributions and structural variations. These issues severely restrict the precise control over polymer chain structure and compromise the controllability and reproducibility of material properties. To solve this problem, this study presents a synergistic catalytic system that combines triethylborane (Et3B), phosphazene base t BuP1 and a thiol initiator to facilitate efficient and controlled anionic ring-opening polymerization of propylene sulfide. Experimental results show that adding Et3B at 0 ℃ effectively blocks the chain transfer reaction of sulfur anions to monomers and prevents the formation of disulfide bonds. Density functional theory calculations confirm that Et3B stabilizes the active sulfur-centered anion intermediates through strong B-S coordination, significantly reducing their nucleophilic reactivity and enabling precise control over the polymerization process. Based on these findings, a “one-pot, two-step” strategy for efficient synthesis of sulfur-rich polymers is developed. By using the dual-component catalytic system, an alternating copolymer of carbon disulfide and propylene sulfide can be synthesized as a macro-chain transfer agent, followed by the direct addition of styrene monomer to successfully create a well-defined polystyrene-co-poly(carbon disulfide-alt-propylene sulfide) terpolymer without isolating the intermediate. This work presents a novel pathway for the precise synthesis of sulfur-rich polymers.
ZHANG Hongxin , ZHU Hongxuan , SUN Duzheng , WANG Guohu , LIU Fengzhuang . Controlled Anionic Polymerization of Propylene Sulfide and Strategy for Synthesis of Sulfur-Rich Copolymers[J]. Journal of South China University of Technology(Natural Science), 2025 , 53(11) : 141 -149 . DOI: 10.12141/j.issn.1000-565X.250115
| [1] | PENCZEK S, CYPRYK M, PRETULA J,et al .Elemental sulfur and cyclic sulfides. Homo- and copolymerizations.Kinetics,thermodynamics and DFT analysis[J].Progress in Polymer Science,2024,152:101818/1-32. |
| [2] | MUTLU H, CEPER E B, LI X,et al .Sulfur chemistry in polymer and materials science[J].Macromolecular Rapid Communications,2019,40(1):1800650/1-51. |
| [3] | YUE T, REN W, LU X .Copolymerization involving sulfur-containing monomers[J].Chemical Reviews,2023,123(24):14038-14083. |
| [4] | IIMORI H, SHIBASAKI Y, ANDO S,et al .Nonstoichiometric polycondensation I. Synthesis of polythioether from dibromomethane and 4,4′‐thiobisbenzenethiol[J].Macromolecular Symposia,2003,199(1):23-36. |
| [5] | CLANCY S, SHRIVER D F, OCHRYMOWYCZ L A .Preparation and characterization of polymeric solid electrolytes from poly(alkylene sulfides) and silver salts[J].Macromolecules,1986,19(3):606-611. |
| [6] | DAGLAR O, ?AKMAK?I E, HIZAL G,et al .Extremely fast synthesis of polythioether based phase change materials (PCMs) for thermal energy storage[J].European Polymer Journal,2020,130:109681/1-11. |
| [7] | ELEMM E, SENSFU? S .Untersuchungen zum selbstinitiierungs‐mechanismus der thiol/En‐polymerisation[J].Die Makromolekulare Chemie,1991,192(1):159-164. |
| [8] | EVANS R A, RIZZARDO E .Free-radical ring-opening polymerization of cyclic allylic sulfides[J].Macromole-cules,1996,29(22):6983-6989. |
| [9] | GAO T, XIA X, TAJIMA K,et al .Polyether/polythioether synthesis via ring-opening polymerization of epoxides and episulfides catalyzed by alkali metal carboxylates[J].Macromolecules,2022,55(21):9373-9383. |
| [10] | LIU Y, CHEN Y, ZHANG Y,et al .Density-adjustable bio-based polysulfide composite prepared by inverse vulcanization and bio-based fillers[J].Polymers,2020,12(9):2127/1-15. |
| [11] | GE C, ZHU J, WU G,et al .ROS-responsive selenopolypeptide micelles:preparation,characterization,and controlled drug release[J].Biomacromolecules,2022,23(6):2647-2654. |
| [12] | YANG W, ZHOU W, DING N,et al .Biobased photothermal responsive shape memory polythioether/MXene nanocomposites with self-extinguishing performance[J].Chemical Engineering Journal,2024,497:154591/1-13. |
| [13] | WANG C, LIANG J, ZHAO Y,et al .All-solid-state lithium batteries enabled by sulfide electrolytes:from fundamental research to practical engineering design[J].Energy & Environmental Science,2021,14(5):2577-2619. |
| [14] | KAMBER N E, JEONE W, WAYMOUTH R M,et al .Organocatalytic ring-opening polymerization[J].Chemical Reviews,2007,107(12):5813-5840. |
| [15] | SONG Q, PASCOUAU C, ZHAO J,et al .Ring-opening polymerization of γ-lactones and copolymerization with other cyclic monomers[J].Progress in Polymer Science,2020,110:101309/1-27. |
| [16] | KIESEWETTER M K, SHIN E J, HEDRICK J L,et al .Organocatalysis: opportunities and challenges for polymer synthesis[J].Macromolecules,2010,43(5):2093-2107. |
| [17] | ZHANG C, ZHANG X .Chemoselective coupling of CS2 and epoxides for producing poly(thioether)s and COS via oxygen/sulfur atom exchange[J].Macromole-cules,2020,53(1):233-239. |
| [18] | YANG H, HUANG J, SONG Y,et al .Anionic hybrid copolymerization of sulfur with acrylate: strategy for synthesis of high-performance sulfur-based polymers[J].Journal of the American Chemical Society,2023,145(26):14539-14547. |
| [19] | CHEN Y, LIU S, ZHAO J,et al .Chemoselective polymerization of epoxides from carboxylic acids: direct access to esterified polyethers and biodegradable polyurethanes[J].ACS Macro Letters,2019,8(12):1582-1587. |
| [20] | XIA Y, SCHEUTZ G M, EASTERLING C P,et al .Hybrid block copolymer synthesis by merging photoiniferter and organocatalytic ring‐opening polymerizations[J].Angewandte Chemie International Edition,2021,60(34):18537-18541. |
| [21] | HONG X, LIU S, PANG J,et al .Polyglycidamides:from backbone‐promoted amidation to degradable polyether with wide‐range LCST[J].Angewandte Chemie International Edition,2025,64(7):e202419978/1-10. |
| [22] | YANG J, WU H, LI Y,et al .Perfectly alternating and regioselective copolymerization of carbonyl sulfide and epoxides by metal‐free Lewis pairs[J].Angewandte Chemie International Edition,2017,56(21):5774-5779. |
| [23] | LUYER S L, GUéGAN P, ILLY N .Episulfide anionic ring-opening polymerization initiated by alcohols and primary amines in the presence of γ-thiolactones[J].Macromolecules,2022,55(13):5430-5440. |
| [24] | CHEN Y, SHEN J, LIU S,et al .High efficiency organic Lewis pair catalyst for ring-opening polymerization of epoxides with chemoselectivity[J].Macromolecules,2018,51(20):8286-8297. |
| [25] | ZHANG D, FENG X, GNANOU Y,et al .Theoretical mechanistic investigation into metal-free alternating copolymerization of CO2 and epoxides: the key role of triethylborane[J].Macromolecules,2018,51(15):5600-5607. |
| [26] | LIU L, ZHAO J, ZHANG G .Chemoselectivity streamlines the approach to linear and Y-shaped thiol-polyethers starting from thiocarboxylic acids[J].ACS Macro Letters,2023,12(9):1185-1192. |
| [27] | NAKANO K, TATSUMI G, NOZAKI K .Synthesis of sulfur-rich polymers: copolymerization of episulfide with carbon disulfide by using[PPN]Cl/(salph)Cr(Ⅲ)Cl System[J].Journal of the American Chemical Society,2007,129(49):15116-15117. |
| [28] | ZHANG C, YANG Y, HE J .Direct transformation of living anionic polymerization into RAFT-based polymerization[J].Macromolecules,2013,46(10):3985-3994. |
/
| 〈 |
|
〉 |