

Journal of South China University of Technology(Natural Science) >
Preparation and CO2 Adsorption Performance of Ca-LTA Zeolite Derived From Titanium-Containing Slag
Received date: 2024-08-23
Online published: 2024-10-08
Supported by
the Open Project of State Key Laboratory of Vanadium and Titanium Resources Comprehensive Utilization(2022P4FZG03A);the Basic Research Planning Program of Yunnan Province(202401AT070384)
LTA-type zeolites is considered a highly promising CO2 capture material due to their excellent pore structures and high selectivity for CO2 adsorption. This study presented a green and sustainable synthesis approach using industrial titanium-containing slag waste as a raw material to prepare Na-LTA zeolite precursors. Calcium loading was adjusted via a conventional liquid-phase ion exchange (LPIE) method to produce a series of xCa-LTA zeolites, specifically designed to enhance CO2 adsorption performance. The adsorption properties of the xCa-LTA zeolites were systematically evaluated through dynamic adsorption experiments. Results show that xCa-LTA zeolites not only significantly enhances CO2 capture capacity, but also exhibits excellent selectivity in CO2/N2 and CO2/CH4 separation processes, with the 0.05Ca-LTA sample demonstrating the most outstanding adsorption performance. Under conditions of 25 ℃ and 105 Pa, the CO2 adsorption rate of 0.05Ca-LTA is 4.95 times that of Na-LTA, with a maximum adsorption capacity of 4.02 mmol/g. Kinetic analysis indicates that the CO2 adsorption behavior of 0.05Ca-LTA follows a pseudo-second-order kinetic model, where the adsorption process is synergistically dominated by both physisorption and chemisorption. This synergy not only accelerated adsorption rates but also improved overall efficiency. After five adsorption/desorption cycles, 0.05Ca-LTA maintains highly efficient and stable adsorption performance, demonstrating excellent cyclic regeneration capability. This study follows the ecofriendly concept of “treating waste with waste” providing a new approach for the high-value utilization of solid waste while offering important theoretical and application potential for the synergistic optimization of CO2 capture and environmental pollution control.
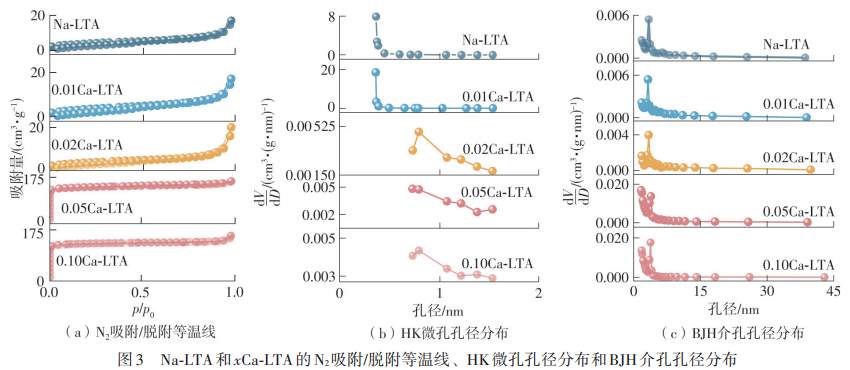
HUANGFU Lin , HE Zhengqing , ZHAO Shimin , ZHOU Xintao , LUO Zhongqiu , ZU Yun , SHANG Bo , LI Fangyuan . Preparation and CO2 Adsorption Performance of Ca-LTA Zeolite Derived From Titanium-Containing Slag[J]. Journal of South China University of Technology(Natural Science), 2025 , 53(3) : 139 -148 . DOI: 10.12141/j.issn.1000-565X.240417
| 1 | 蒲龙映,彭青松,李乐军 .含钛高炉渣综合利用研究现状与展望[J].环境生态学,2022,4(2/3):106-110. |
| PU Long-ying, PENG Qing-song, LI Le-jun .Research status and new technology of comprehensive utilization of titanium-bearing blast furnace slag[J].Environmental Ecology,2022,4(2/3):106-110. | |
| 2 | 宋洋,曾荣,陶从喜,等 .钛矿渣作水泥混合材的应用研究[J].水泥技术,2022(2):68-73. |
| SONG Yang, ZENG Rong, TAO Congxi,et al .Study on application of titanium slag as cement mixture[J].Cement Technology,2022(2):68-73. | |
| 3 | 王帅,吕淑珍,赵杰,等 .高钛矿渣制备混凝土用矿物掺合料研究[J].西南科技大学学报,2021,36(1):28-34. |
| WANG Shuai, Shuzhen Lü, ZHAO Jie,et al .Preparation of mineral admixture for concrete with high titanium slag[J].Journal of Southwest University of Science and Technology,2021,36(1):28-34. | |
| 4 | 许莹,张晓蒙,施展 .含钛高炉渣制备免烧免蒸砖的研究[J].新型建筑材料,2015,42(8):53-55. |
| XU Ying, ZHANG Xiaomeng, SHI Zhan .Study on non-autoclaved and unburnt bricks prepared by Ti-bearing blast furnace slag[J].New Building Materials,2015,42(8):53-55. | |
| 5 | 尤皓,孙红娟,彭同江 .烧结温度对提钛渣微晶玻璃析晶及显微结构的影响[J].材料科学与工程学报,2020,38(3):450-454,465. |
| YOU Hao, SUN Hongjuan, PENG Tongjiang .Effects of sintering temperature on crystallization and microstructure of glass ceramics from slags after extracting titanium[J].Journal of Materials Science & Engineering,2020,38(3):450-454,465. | |
| 6 | LI X Y, LI J, LU Z Y,et al .Preparation and properties of reactive powder concrete by using titanium slag aggregates[J].Construction and Building Materials,2020,234:117342/1-14. |
| 7 | 梁止水,高琦,刘豪伟,等 .煤矸石制备NaX型分子筛及其对Cd2+的吸附性能[J].东南大学学报(自然科学版),2020,50(4):741-747. |
| LIANG Zhishui, GAO Qi, LIU Haowei,et al .Synthesis of NaX zeolite from coal gangue and its adsorption capability for Cd2+ [J].Journal of Southeast University (Natural Science Edition),2020,50(4):741-747. | |
| 8 | CHEN Z, LI X G, LIU H H,et al .A novel and cost-effective synthesis of magnetic zeolite 4A using kaolinite and red mud for Sr(Ⅱ) removal[J].Microporous and Mesoporous Materials,2024,370:113069/1-10. |
| 9 | CHEN W T, SONG G Q, LIN Y Y,et al .A green and efficient strategy for utilizing of coal fly ash to synthesize K-MER zeolite as catalyst for cyanoethylation and adsorbent of CO2 [J].Microporous and Mesoporous Materials,2021,326:111353/1-10. |
| 10 | KALVACHEV Y, ZGUREVA D, BOYCHEVA S,et al .Synthesis of carbon dioxide adsorbents by zeolitization of fly ash[J].Journal of Thermal Analysis and Calorimetry,2016,124:101-106. |
| 11 | VERRECCHIA G, CAFIERO L, DE CAPRARIIS B,et al .Study of the parameters of zeolites synthesis from coal fly ash in order to optimize their CO2 adsorption [J].Fuel,2020,276:118041/1-10. |
| 12 | 皇甫林,尚波,罗中秋,等 .含钛渣制备多孔沸石分子筛吸附剂及其CO2吸附性能[J].精细化工,2024,41(8):1726-1736,1856. |
| HUANGFU Lin, SHANG Bo, LUO Zhongqiu,et al .Preparation and CO2 adsorption performance of porous zeolite molecular sieve adsorbents using titanium-bearing slag [J].Fine Chemicals,2024,41(8):1726-1736,1856. | |
| 13 | YANG K Z, YANG G, WU J Y,et al .Insights into the enhancement of CO2 adsorption on faujasite with a low Si/Al ratio:understanding the formation sequence of adsorption complexes[J].Chemical Engineering Journal,2021,404:127056/1-10. |
| 14 | BOER D G, LANGERAK J, PESCARMONA P P .Zeolites as selective adsorbents for CO2 separation[J].ACS Applied Energy Materials,2023,6(5):2634-2656. |
| 15 | KRISHNA R .Diffusion in porous crystalline materials[J].Chemical Society Reviews,2012,41(8):3099-3118. |
| 16 | WANG B H, ZHU Y, QIN Q Y,et al .Development on hydrophobic modification of aluminosilicate and titanosilicate zeolite molecular sieves[J].Applied Catalysis A:General,2021,611:117952/1-17. |
| 17 | ZHANG C, ZHANG D Y, ZHANG X Q,et al .Rubik’s cube-shaped Linde type A zeolite synthesized from biomass for carbon dioxide capture[J].Cell Reports Physical Science,2024,5(4):101889/1-17. |
| 18 | TAO Z Y, TIAN Y M, OU S Y,et al .Direct air capture of CO2 by metal cation-exchanged LTA zeolites:effect of the charge-to-size ratio of cations[J].AIChE Journal, 2023,69(8):e18139/1-11. |
| 19 | ZHU H, LI S M, ZHANG J F,et al .A highly effective and low-cost sepiolite-based solid amine adsorbent for CO2 capture in post-combustion[J].Separation and Purification Technology,2023,306:122627/1-11. |
| 20 | CHOI H J, HONG S B .Effect of framework Si/Al ratio on the mechanism of CO2 adsorption on the small-pore zeolite gismondine[J].Chemical Engineering Journal,2022,433:133800/1-9. |
| 21 | QU F, YAN F, SHEN X,et al .Novel PEI@CSH adsorbents derived from coal fly ash enabling efficient and in-situ CO2 capture: the anti-urea mechanism of CSH support [J].Journal of Cleaner Production,2022,378:134420/1-10. |
| 22 | STARKE A, PASEL C, BL?KER C,et al .Impact of Na+ and Ca2+ cations on the adsorption of H2S on binder-free LTA zeolites[J].Adsorption Science & Technology,2021,2021:5531974/1-12. |
| 23 | SUN X Y, ZHANG Q Q, LI S H,et al .Maximizing the utilization of calcium species in the supercages of CaNa-FAU zeolite for efficient CO2 capture[J].Chemical Engineering Journal,2024,481:148661/1-14. |
| 24 | LOIOLA A R, ANDRADE J C R A, SASAKI J M,et al .Structural analysis of zeolite NaA synthesized by a cost-effective hydrothermal method using kaolin and its use as water softener[J].Journal of Colloid and Interface Science,2012,367(1):34-39. |
| 25 | SINGH J, WHITE R L .A variable temperature infrared spectroscopy study of CaA zeolite dehydration and carbonate formation[J].Spectrochimica Acta Part A:Molecular and Biomolecular Spectroscopy,2019,207:189-196. |
| 26 | CUI Y K, XING Y, TIAN J L,et al .Insights into the adsorption performance and separation mechanisms for CO2 and CO on NaX and CaA zeolites by experiments and simulation[J].Fuel,2023,337:127179/1-10. |
| 27 | DRENCHEV N L, IVANOVA E Z, MIHAYLOV M Y,et al .One Ca2+ site in CaNaY zeolite can attach three CO2 molecules[J].The Journal of Physical Chemistry Letters,2023,14(6):1564-1569. |
| 28 | BAE T H, HUDSON M R, MASON J A,et al .Evaluation of cation-exchanged zeolite adsorbents for post-combustion carbon dioxide capture[J].Energy & Environmental Science,2013,6:128-138. |
| 29 | CHEN C, AHN W S .CO2 adsorption on LTA zeolites:effect of mesoporosity[J].Applied Surface Science,2014,311:107-109. |
| 30 | KUMAR S, SRIVASTAVA R,KOH J .Utilization of zeolites as CO2 capturing agents:advances and future perspectives[J].Journal of CO2 Utilization,2020,41:101251/1-16. |
/
| 〈 |
|
〉 |