

Journal of South China University of Technology(Natural Science) >
Dissociation Behavior of Polyacrylic Acid in Aqueous Solution
Received date: 2022-06-20
Online published: 2022-07-18
Supported by
the National Natural Science Foundation of China(52073099)
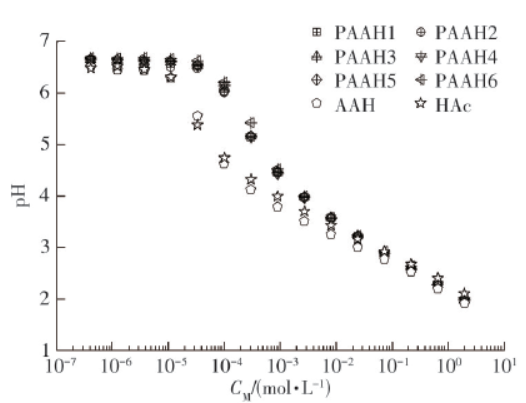
In order to study the dissociation behavior of polyacrylic acid (PAAH) with different molecular weight in aqueous solution as well as the difference between the dissociation behavior of polyelectrolyte (polyacrylic acid) and small molecular weak electrolyte (acrylic acid (AAH) and acetic acid (HAc)), this paper prepared polyacrylic acid with polymerization degree in the range of 300~2 000 by precipitation polymerization method. The pH value of polyacrylic acid, acrylic acid and acetic acid aqueous solution was measured by pH meter to determine the dissociation behavior of electrolyte. Then from the thermodynamic point of view and Flory-Huggins lattice model, the paper derived the dissociation equilibrium equation of electrolyte in the dissociation process. The results show that the dissociation behavior of polyacrylic acid in aqueous solution is independent of the degree of polymerization. By simplifying the dissociation equilibrium equation, the calculation formula of the traditional equilibrium constant Kα was obtained and given its exact physical meaning: the relative strength parameter of the interaction energy of the dissociation hydration effect of electrolyte aqueous solution in the dissociation process. By substituting the data into the dissociation equilibrium equation, it is found that there are two kinds of molar ionization hydration energies ∆Es in polyelectrolyte and small molecule weak electrolyte, in the low concentration region and the high concentration region, respectively. This is the reason why acrylic acid, acetic acid and polyacrylic acid deviate from the traditional equilibrium constant Kα theoretical line in the low concentration region.
LI Wenbo , DU Xiaoxiao . Dissociation Behavior of Polyacrylic Acid in Aqueous Solution[J]. Journal of South China University of Technology(Natural Science), 2023 , 51(3) : 83 -90 . DOI: 10.12141/j.issn.1000-565X.220385
| 1 | BORUKHOV I, ANDELMAN D, BORREGA R,et al .Polyelectrolyte titration:Theory and experiment [J].Journal of Physical Chemistry B,2000,104(47):11027-11034. |
| 2 | CARRILLO J M, DOBRYNIN A V .Salt effect on osmotic pressure of polyelectrolyte solutions:Simulation study [J].Polymers,2014,6(7):1897-1913. |
| 3 | LANDSGESELL J, NOVA L,RUD O,et al .Simulations of ionization equilibria in weak polyelectrolyte solutions and gels [J].Soft Matter,2019,15(6):1155-1185. |
| 4 | WANDREY C .Concentration regimes in polyelectrolyte solutions [J].Langmuir,1999,15(12):4069-4075. |
| 5 | XU G F, LUO S J, YANG Q B,et al .Single chains of strong polyelectrolytes in aqueous solutions at extreme dilution:Conformation and counterion distribution[J].Journal of Chemistry Physics,2016,145(14):144903-1-7. |
| 6 | 刘全伟,李艺,王治流,等 .丙烯酸-丙烯酸钠共聚物在水溶液中的离解 [J].高分子学报,2005(4):484-485. |
| LIU Quan-wei, LI Yi, WANG Zhi-liu,et al .Dissociation of acrylic acid sodium acrylate copolymer in aqueous solution [J].Acta Polymerica Sinica,2005(4):484-485. | |
| 7 | FUKUSHIMA M, TATSUMI K, WADA S .Evaluation of the intrinsic acid-dissociation constant of alginic acid by considering the electrostatic effect [J].Analytical Sciences,1999,15:1153-1155. |
| 8 | YEKYMOV E, ATTIA D, LEVI-KALISMAN Y,et al .Effects of non-ionic micelles on the acid-base equilibria of a weak polyelectrolyte [J].Polymers,2022,14(9):1926. |
| 9 | PAN Y, CHENG R S .A novel interpretation of concentration dependence of viscosity of dilute polymer solution [J].Chinese Journal of Poylmer Science,2000,18(1):57-67. |
| 10 | KAJI K, URAKAWA H, KANAYA T,et al .Phase diagram of polyelectrolyte solutions [J].Journal de Physique France,1988,49(6):993-1000. |
| 11 | TANAHATOE J J, KUIL M E .Dynamic light scattering of a flexible highly charged polyelectrolyte in the dilute concentration regime [J].Macromolecules,1997,30(20):6102-6106. |
| 12 | 蔡佳利,薄淑琴,程镕时 .聚电解质溶液浓度区域的划分 [J].高分子学报,2004(5):625-627. |
| Cai Jia-li, Bo Shu-qin, Cheng Rong-shi .Division of concentration region of polyelectrolyte solution [J].Acta Polymerica Sinica,2004(5):625-627. | |
| 13 | NISHIDA K, KAJI K, KANAYA T .Improved phase diagram of polyelectrolyte solutions [J].Journal of Chemical Physics,2001,115(17):8217-8220. |
| 14 | NISHIDA K, KAJI K, KANAYA T .High concentration crossovers of polyelectrolyte solutions [J].Journal of Physical Chemistry,2001,114(19):8671-8677. |
| 15 | WANDREY C .Concentration regimes in polyelectrolyte solutions [J].Langmuir,1999,15(12):4069-4075. |
| 16 | MUTHUKUMAR M .Theory of counter-ion condensation on flexible polyelectrolytes:Adsorption mechanism [J].Journal of Chemical Physics,2004,120(19):9343-9350. |
| 17 | 蔡正春,杨琥,程镕时 .聚电解质在水溶液中的离解行为[C]∥2007年全国高分子学术论文报告会论文摘要集(上册).成都:[s.n.],2007:237. |
| 18 | CHENG R S .On the concentration regimes of a flexible-chain polymer solution [J].Macromolecular Symposia,1997,124:27-34. |
| 19 | 蔡正春 .聚丙烯酸在水溶液中的离解与电迁移行为 [D].南京:南京大学,2008. |
| 20 | 陈环环 .聚电解质在溶液中的电离行为 [D].广州:华南理工大学,2016. |
| 21 | 蔡正春,杨琥,程镕时 .羧酸离子在水中的水合数 [J].化学学报,2008,66(7):831-833. |
| CAI Zheng-chun, YANG Hu, CHENG Rong-shi .Hydration number of carboxylic acid ions in water [J].Acta Chimica Sinica,2008,66(7):831-833. | |
| 22 | LI W B, ZHENG Y, CHENG R S .Transition of hydration states of poly(vinyl alcohol) in aqueous solution [J].Polymer,2008,49(21):4740-4744. |
| 23 | LI W B, XUE F, CHENG R S .States of water in partially swollen poly(vinyl alcohol) hydrogels [J].Polymer,2005,46(25):12026-12031. |
/
| 〈 |
|
〉 |