

Journal of South China University of Technology(Natural Science) >
Analysis of Adsorption Performance of 13X Molecular Sieve for Low Concentration C3F8
Received date: 2022-03-14
Online published: 2022-05-13
Supported by
the National Natural Science Foundation of China(22075089)
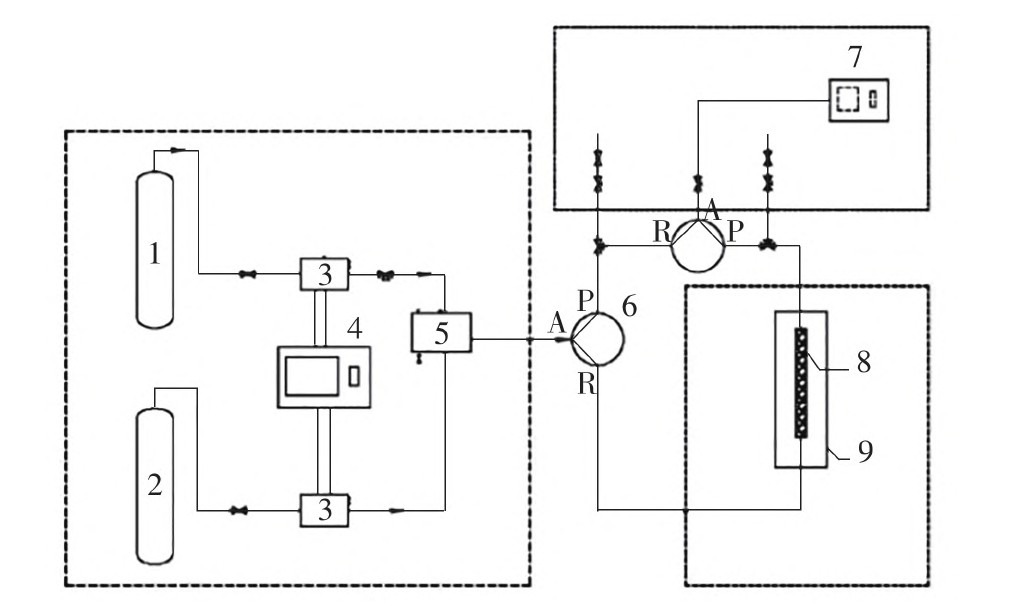
The presence of perfluoropropane (C3F8) in the electronics industry will affect the arcing and insulation performance of sulfur hexafluoride (SF6) gas, it is necessary to separate C3F8. However, at present, C3F8 of low concentration can not be removed through cryoscopic separation method, and the research reports about using adsorption separation method for the adsorption of C3F8 are still few. This paper used the commercial 13X molecular sieve for adsorption of low concentration C3F8. As well, it simulated the industrial fisxed bed adsorption experiment and measured the dynamic adsorption performance of C3F8 on 13X molecular sieve. The breakthrough curve of commercial 13X molecular sieve for low concentration of C3F8 (ppm) was studied with the change of inlet flow rate, inlet C3F8 mass concentration and adsorption temperature, and the influence degree of each factor was analyzed. The adsorption equilibrium model and kinetic model were fitted by adsorption transmission curve to analyze the adsorption behavior and mechanism of 13X molecular sieve for low concentration of C3F8. The results show that the influence of these three factors orders as follows: inlet gas mass concentration > temperature > inlet flow rate. The adsorption equilibrium model accords with the Langmuir isothermal adsorption model, and the kinetic adsorption model accords with the pseudo-first-order kinetic model. When the temperature is 30 ℃, the inlet flow rate is 1.2 L/min, the pressure is 0.12 MPa, and the mass concentration of C3F8 gas is 459 mg/L (ppm), the dynamic adsorption of C3F8 by 13X molecular sieve can reach 5.428 mmol/g, which provides data reference for the industrial adsorption process of C3F8.
YUAN Wenhui, YE Zhaochun, LI Li, et al . Analysis of Adsorption Performance of 13X Molecular Sieve for Low Concentration C3F8[J]. Journal of South China University of Technology(Natural Science), 2023 , 51(3) : 74 -82 . DOI: 10.12141/j.issn.1000-565X.220118
| 1 | 张亮 .电力设备中SF6混合绝缘气体的净化分离技术[J].黑龙江科学,2021,12(20):84-85. |
| ZHANG Liang .Purification separation technology of SF6 mixture insulating gas in the electrical equipment[J].Heilongjiang Science,2021,12(20):84-85. | |
| 2 | 廖恒易 .浅谈气体的分离和提纯[J].低温与特气,2015,33(6):5-7. |
| LIAO Hengyi .Gas separation and purification[J].Low Temperature and Specialty Gases,2015,33(6):5-7. | |
| 3 | 刘伟,苏镇西,祁炯,等 .电力设备中SF6混合绝缘气体净化分离技术的探究[J].高压电器,2016,52(12):227-231. |
| LIU Wei, SU Zhenxi, QI Jiong,et al .Investigation into the purification and separation technology for SF6 gas mixtures used in electrical apparatus[J].High Voltage Apparatus,2016,52(12):227-231. | |
| 4 | Lü D F, LIU Z W, XU F,et al .A Ni-based metal-organic framework with super-high C3H8 uptake for adsorptive separation of light alkanes[J].Separation and Purification Technology,2021,266:118198-1-10. |
| 5 | DU X D, PANG D D, CHENG Y G,et al .Adsorption of CH4,N2,CO2,and their mixture on montmorillonite with implications for enhanced hydrocarbon extraction by gas injection[J].Applied Clay Science,2021,210:106160-1-11. |
| 6 | WEBLEY P A .Adsorption technology for CO2 separation and capture:A perspective[J].Adsorption,2014,20(2/3):225-231. |
| 7 | ???,???, YOO Y D,et al .Analysis of CO2 adsorption characteristic of differently manufactured zeolite 13X[J].Journal of Energy & Climate Change,2008,3(1):34-40. |
| 8 | LAMIA N, WOLFF L, LEFLAIVE P,et al .Propane/propylene separation by simulated moving bed I.Adsorption of propane,propylene and isobutane in pellets of 13X zeolite[J].Separation Science and Technology,2007,42(12):2539-2566. |
| 9 | de OLIVEIRA L H, MENEGUIN J G, PEREIRA M V,et al .H2S adsorption on NaY zeolite[J].Microporous and Mesoporous Materials,2019,284:247-257. |
| 10 | 胡苏阳,刘鑫博,唐建峰,等 .13X沸石分子筛对低浓度CO2动态吸附[J].化工进展,2022,41(1):153-160. |
| HU Suyang, LIU Xinbo, TANG Jianfeng,et al .Dynamic adsorption of low concentration CO2 over 13X zeolite[J].Chemical Industry and Engineering Progress,2022,41(1):153-160. | |
| 11 | HONG T Q, WEI L, CUI K P,et al .Adsorption performance of volatile organic compounds on activated carbon fibers in a fixed bed column[J].Journal of Environmental Chemical Engineering,2021,9(6):106347-1-8. |
| 12 | GUO Z H, SUN Z N, ZHANG N,et al .Mean porosity variations in packed bed of monosized spheres with small tube-to-particle diameter ratios[J].Powder Technology,2019,354:842-853. |
| 13 | MCCABE W L S J C, HARRIOTT P .Unit operations of chemical engineering[M].7th ed.New York:McGraw-Hill College,2004:110-133. |
| 14 | 唐进京,鲁军辉,李俊明,等 .CO2/H2O吸附分离特性研究[J].中国电机工程学报,2022,42(6):2216-2227. |
| TANG Jinjing, LU Junhui, LI Junming,et al .Study on adsorption characteristics of CO2/H2O[J].Proceedings of the CSEE,2022,42(6):2216-2227. | |
| 15 | SHENDALMAN L H, MITCHELL J E .A study of heatless adsorption in the model system CO2 in He,I[J].Chemical Engineering Science,1972,27(7):1449-1458. |
| 16 | POURSAEIDESFAHANI A, ANDRES-GARCIA E, de LANGE M,et al .Prediction of adsorption isotherms from breakthrough curves[J].Microporous and Mesoporous Materials,2019,277:237-244. |
| 17 | CRESPO D, QI G, WANG Y,et al .Superior sorbent for natural gas desulfurization[J].Industrial & Engineering Chemistry Research,2008,47(4):1238-1244. |
| 18 | DA N E, SAYARI A .Modeling adsorption of copper on amine-functionalized SBA-15:Predicting breakthrough curves[J].Journal of Environmental Engineering,2013,139(1):95-103. |
| 19 | DOWNAROWICZ D, ALEKSANDRZAK T .Isobutanol vapor adsorption on activated carbons:Equilibrium and kinetic studies[J].Journal of Chemical and Engineering Data,2017,62(10):3518-3524. |
| 20 | HAMOUDI S, SAAD R, BELKACEMI K .Modeling breakthrough curves for adsorption of monobasic phosphate using ammonium-functionalized MCM-48[J].Separation Science and Technology,2013,48(14):2099-2107. |
| 21 | NG M, SCHORK J M, FABREGAS K R .The mass transfer zone in nitrogen PSA columns[J].Gas Separation & Purifications,1993,7(3):159-166. |
| 22 | ZHANG Y H, JIN F, SHEN Z T,et al .Adsorption of methyl tert-butyl ether (MTBE) onto ZSM-5 zeolite:Fixed-bed column tests,breakthrough curve modelling and regeneration[J].Chemosphere,2019,220:422-431. |
| 23 | CHERN J M, CHIEN Y W .Adsorption of nitrophenol onto activated carbon:Isotherms and breakthrough curves[J].Water Research,2002,36(3):647-655. |
| 24 | FOMKIN A A, NIKIFOROV Y V, SINITSYN V A,et al .Adsorption of perfluoropropane on the PAC microporous carbon adsorbent[J].Russian Chemical Bulletin,2002,51(12):2161-2164. |
| 25 | YANG C F O, KAM S H, LIU C H,et al .Assessment of removal efficiency of perfluorocompounds (PFCs) in a semiconductor fabrication plant by gas chromatography[J].Chemosphere,2009,76(9):1273-1277. |
| 26 | LIN B Y, CHANG M B, CHEN H L,et al .Removal of C3F8 via the combination of non-thermal plasma,adsorption and catalysis[J].Plasma Chemistry and Plasma Processing,2011,31(4):585-594. |
| 27 | KOLOKOLOV D I, ARZUMANOV S S, FREUDE D,et al .Mobility of stable π-complexes of ethylene with Ag+ cations in Ag/H-ZSM-5 zeolite:A 2H solid-state NMR study[J].The Journal of Physical Chemistry C,2016,120(9):4993-5000. |
| 28 | ZHANG X, CHEN S, BI H T .Application of wave propagation theory to adsorption breakthrough studies of toluene on activated carbon fiber beds[J].Carbon,2010,48(8):2317-2326. |
| 29 | YI H, YANG X, TANG X,et al .Removal of toluene from industrial gas over 13X zeolite supported catalysts by adsorption-plasma catalytic process[J].Journal of Chemical Technology and Biotechnology,2017,92(9):2276-2286. |
| 30 | CHEN S J, FU Y, HUANG Y X,et al .Experimental investigation of CO2 separation by adsorption methods in natural gas purification[J].Applied Energy,2016,179:329-337. |
| 31 | JAHROMI P E, FATEMI S, VATANI A,et al .Purification of helium from a cryogenic natural gas nitrogen rejection unit by pressure swing adsorption[J].Separation and Purification Technology,2018,193:91-102. |
| 32 | HAJILARI M, SHARIATI A, KHOSRAVI-NIKOU M .Mass transfer determination of ethanol adsorption on activated carbon:Kinetic adsorption modeling[J].Heat and Mass Transfer,2019,55(8):2165-2171. |
/
| 〈 |
|
〉 |