

Journal of South China University of Technology(Natural Science) >
Preparation of PEO-Based Solid Polymer Electrolyte Membranes via Solvent-Free Melting and Properties of the Product
Received date: 2021-12-17
Online published: 2022-03-25
Supported by
the National Natural Science Foundation of Guangdong Province(2021A1515012609)
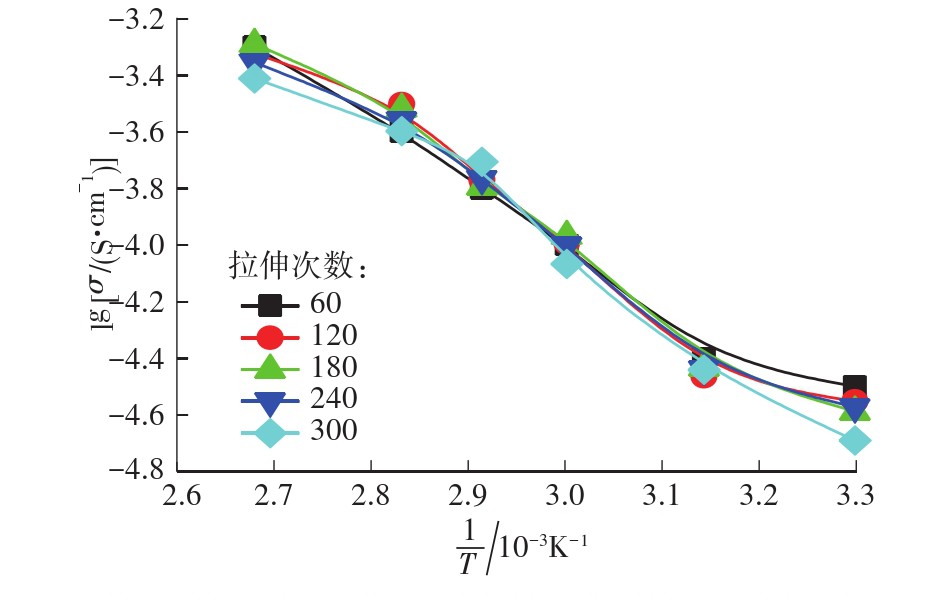
With the advancement of science and technology, various intelligent electronic devices, electric vehicles and grid systems have grown rapidly, and people have a growing demand for high energy density and rechargeable battery systems. In the past few decades, the development of lithium-ion batteries has made great progress. Lithium-ion batteries consisting of positive and negative electrodes, electrolytes and separators has the danger of volatile and leakage, leading to short circuit, fire, explosion and other safety accidents. The emergence of solid electrolytes has largely eliminated this safety hazard. The solid-state electrolyte can not only separate the positive and negative electrodes in the battery to prevent internal short circuits, but also act as an ion conductor to achieve the dual role of lithium ion transfer between positive and negative active substances. The solid-state electrolyte has become one of the research hotspots in the field of new energy. At present, the preparation methods of solid electrolyte membranes mainly include organic solvent casting/coating or hot molding. Organic solvents used by these methods are not friendly to the environment and these methods have disadvantages like high manufacturing cost and low production efficiency. To solve this problem, this paper proposed a new method for preparing PEO-based composite solid electrolyte membrane based on tensile stress and solvent-free melting, characterized the crystalline morpho-logy and microscopic morphology of the electrolyte membrane by SEM and XRD, and characterized the thermal properties of the membrane by DSC and FT-IR. The electrochemical performance of the membrane was characte-rized by linear sweep voltammetry and conductivity test. The results show that, as compared with simple mechanical mixing, the PEO/LiTFSI/PVDF composite solid electrolyte membrane prepared based on tensile stress and solvent-free melting has better lithium salt dispersibility. Ionic conductivity test shows that, even the proposed method does not use organic solvents, it is still comparable to the ionic conductivity of electrolyte membranes prepared by the solution method. When the content of PVDF-HFP is 30%, the ionic conductivity of the PEO-based solid polymer electrolyte membrane at 60 ℃ reaches 2.07×10-4 S/cm, and the electrochemical stability window is 5.0 V.
ZHANG Guizhen LIU Yang YAN Mingbao . Preparation of PEO-Based Solid Polymer Electrolyte Membranes via Solvent-Free Melting and Properties of the Product[J]. Journal of South China University of Technology(Natural Science), 2022 , 50(8) : 119 -127 . DOI: 10.12141/j.issn.1000-565X.210805
| 1 | LIN D C, LIU Y Y, CUI Y .Reviving the lithium metal anode for high-energy batteries[J].Nature Nanotechnology,2017,12(3):194-206. |
| 2 | ZHU M, WU J, WANG Y,et al .Recent advances in gel polymer electrolyte for high-performance lithium ba-tteries[J].Journal of Energy Chemistry,2019,37:126-142. |
| 3 | YUE L, MA J, ZHANG J,et al .All solid-state polymer electrolytes for high-performance lithium ion batteries[J].Energy Storage Materials,2016,5:139-164. |
| 4 | BANDARA L, DDISSANAYAKE M, MELLANDER B E .Ionic conductivity of plasticized (PEO)-LiCF3SO3 electrolytes[J].Electrochimica Acta,1998,43(10/11):1447-1451. |
| 5 | SUN C, LIU J, GONG Y,et al .Recent advances in all-solid-state rechargeable lithium batteries[J].Nano Energy,2017,33:363-386. |
| 6 | 赵嫣然,陈少杰,陶益成,等 .新型PEO/LPOS复合聚合物电解质的制备与性能研究[J].储能科学与技术,2016,5(5):730-734. |
| 6 | ZHAO Yanran, CHEN Shaojie, TAO Yicheng,et al .Preparation and performance of novel PEO/LPOS composite polymer electrolytes[J].Energy Storage Science and Technology,2016,5(5):730-734. |
| 7 | 屠芳芳,谢健,郭锋,等 .基于Li 6.4La3Zr1.4Ta0 .6O12/聚合物复合固态电解质的制备及电化学性能[J].无机化学学报,2020,36(8):1515-1523. |
| 7 | TU Fang-fang, XIE Jian, GUO Feng,et al .Preparation and electrochemical properties of Li6.4La3Zr1.4Ta0.6O12/polymer composite solid electrolyte[J].Acta Inorganic Chimica Sinica,2020,36(8):1515-1523. |
| 8 | NIEDZICKI L, KASPRZYK M, KUZIAK K,et al .Modern generation of polymer electrolytes based on lithium conductive imidazole salts[J].Journal of Power Sources,2009,192(2):612-617. |
| 9 | SCHNELL J, GUENTHER T, KNOCHE T,et al .All-solid-state lithium-ion and lithium metal batteries-paving the way to large-scale production[J].Journal of Power Sources,2018,382:160-175. |
| 10 | ZEKOLL S, MARRINER-EDWARDS C, HEKSELMAN A K O,et al .Hybrid electrolytes with 3D bicontinuous ordered ceramic and polymer microchannels for all-solid-state batteries[J].Energy & Environmental Science,2018,11(1):185-201. |
| 11 | PORCARELLI L, GERBALDI C, BELLA F,et al .Super soft all-ethylene oxide polymer electrolyte for safe all-solid lithium batteries[J].Scientific Reports,2016,6(1):1-14. |
| 12 | GRAY F M, MACCALLUM J R, VINCENT C A .Poly(ethylene oxide)-LiCF3SO3-polystyrene electrolyte systems[J].Solid State Ionics,Diffusion & Reactions,1986,18-19:282-286. |
| 13 | 马文有,田秋,曹茂盛,等 .纳米颗粒分散技术研究进展——分散方法与机理(1)[J].中国粉体技术,2002,8(3):28-31. |
| 13 | MA Wen-you, TIAN Qiu, CAO Mao-sheng,et al .Research progress on nanoparticle dispersion technolo-gy——dispersion methods and mechanisms (1)[J].China Powder Technology,2002,8(3):28-31. |
| 14 | CHEN L, LI Y, LI S P,et al .PEO/garnet composite electrolytes for solid-state lithium batteries:from “ceramic-in-polymer”to“polymer-in-ceramic”[J].Nano Energy,2018,46:176-184. |
| 15 | 瞿金平,张桂珍,殷小春 .一种偏心转子体积脉动形变塑化输运装置:201410206552.8[P].2014-10-08. |
| 16 | WU T, YUAN D, QIU F,et al .Polypropylene/polystyrene/clay blends prepared by an innovative eccentric rotor extruder based on continuous elongational flow:analysis of morphology,rheology property,and crystallization behavior[J].Polymer Testing,2017,63:73-83. |
| 17 | ZHANG G Z, WU T, LIN W Y,et al .Preparation of polymer/clay nanocomposites via melt intercalation under continuous elongation flow[J].Composites Science and Technology,2017,145:157-164. |
/
| 〈 |
|
〉 |