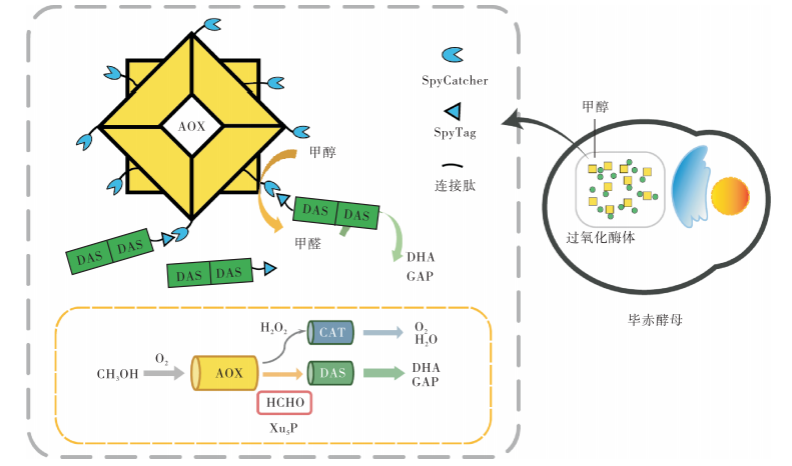
As a kind of methanol-trophic yeast, Pichia pastoris can use methanol as the only carbon source and energy source, and it is one of the widely used expression systems for production of eukaryotic exogenous proteins. However, the utilization and assimilation efficiency of methanol in Pichia pastoris is low, and the methanol of high concentration is toxic to the cell. In the process of metabolizing methanol, the accumulate toxic intermediate metabo-lites such as formaldehyde and formic acid will severely repress cell growth, and limit the synthesis of target proteins.This study designed to construct a substrate channel between the key enzymes in methanol metabolism with multi-enzyme-assembly technology, aiming to reduce the accumulation and diffusion of formaldehyde, improve the flux of the assimilation pathway, enhance the tolerability of Pichia pastoris, and improve Pichia pastoris ability to express foreign protein. Thus Pichia pastoris artificial cell with high methanol utilization can be obtained. In this paper, the feasibility of the multi-enzyme assembly technology—the SpyTag/SpyCatcher system in Pichia pastoris was firstly identified by the bimolecular fluorescence complementation (BiFC) analysis technology. Then alcohol oxidase (AOX) and dihydroxyacetone synthase (DAS) were assembled to form a self-assemble double-enzyme complex with SpyTag and SpyCatcher. On the basis of that, the green fluorescent protein (EGFP) was introduced into the recombinant strain as a reporter protein to analyze the comsumption of methanol for cell growth and the production of recombinant protein. The results show that, when 2% methanol is used as the carbon source, the biomass of the recombinant strain expressing the double-enzyme self-assembly complex is 2.3 times bigger than that of the original strain, and its EGFP fluorescence intensity of per D(600) cell is 4.51 times higher than that of the original strain, and it is 2.76 times of the unassembled strains EGFP fluorescence intensity;the production of EGFP is about 1.52 mg/mL.
HAN Shuangyan LI Jingwen WANG Yuanyuan
. Production Capacity Improvement of Recombinant Proteins of Pichia pastoris with the SpyTag/SpyCatcher System[J]. Journal of South China University of Technology(Natural Science), 2022
, 50(1)
: 143
-154
.
DOI: 10.12141/j.issn.1000-565X.210359


