

Journal of South China University of Technology(Natural Science) >
Recombinant Expression and Biochemical Characterization of Unspecific Peroxygenase GmaUPO
Received date: 2021-01-29
Revised date: 2021-03-09
Online published: 2021-03-21
Supported by
Supported by the National Natural Science Foundation of China(31930084,31725022,32001633)
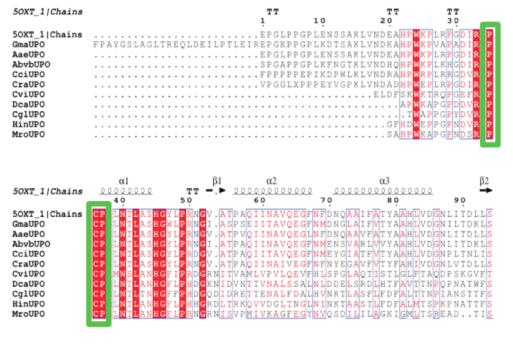
Key words: unspecific peroxygenase; Galerina marginata; cloning; expression; Pichia pastoris
LAN Dongming NIE Xiaoshuang MA Yunjian WANG Yonghua . Recombinant Expression and Biochemical Characterization of Unspecific Peroxygenase GmaUPO[J]. Journal of South China University of Technology(Natural Science), 2021 , 49(9) : 22 -28 . DOI: 10.12141/j.issn.1000-565X.210050
/
| 〈 |
|
〉 |