

收稿日期: 2025-08-27
网络出版日期: 2025-09-15
基金资助
国家重点研发计划项目(2024YFF1106803)
Effect of Chicken Peptide-Ferrous Chelate on Iron Deficiency Anemia in Mice
Received date: 2025-08-27
Online published: 2025-09-15
Supported by
the Research and Development Program of China(2024YFF1106803)
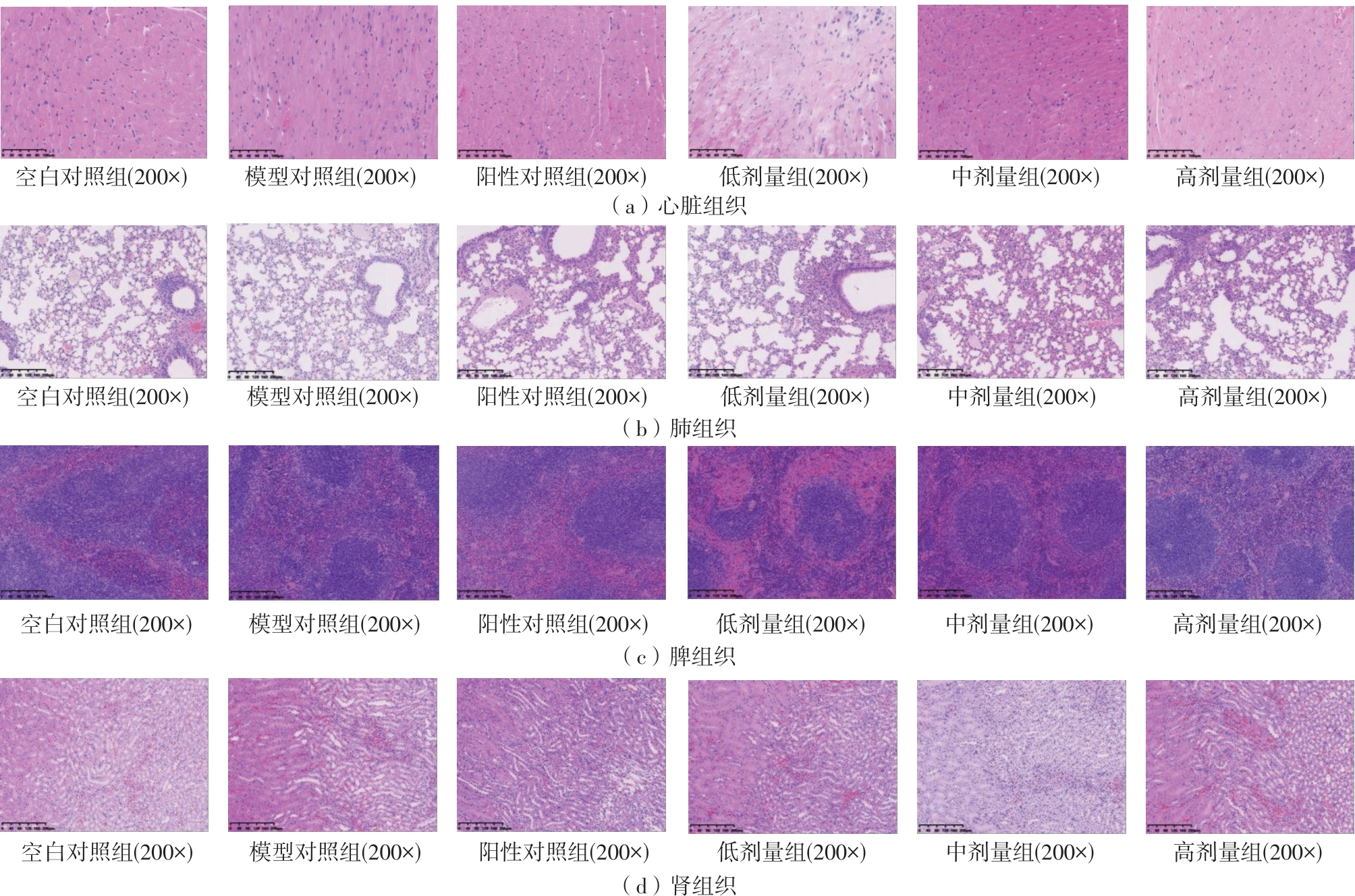
该文系统探讨了鸡肉肽-亚铁螯合物(CMP-Fe)对缺铁性贫血(IDA)小鼠贫血症状的改善作用,并从体质量变化、血常规参数、铁代谢指标、炎症反应及组织病理学等多个维度,综合评价其干预效应。实验结果表明:与模型组相比,CMP-Fe各剂量组(按小鼠体质量计,小、中、高剂量组分别喂食Fe 1.0、2.0、3.0 mg/kg)的小鼠体质量、血常规指标(包括红细胞计数RBC、血红蛋白HGB、红细胞压积HCT、平均红细胞体积MCV、平均红细胞血红蛋白含量MCH、平均红细胞血红蛋白浓度MCHC、红细胞分布宽度-变异系数RDW-CV)及血清铁代谢指标(包括血清铁SI、总铁结合力TIBC、转铁蛋白TRF、铁蛋白FER、转铁蛋白饱和度TSAT、不饱和铁结合力UIBC)均得到显著改善,且高剂量组效果最为突出。CMP-Fe对RBC、HGB、HCT等红细胞系指标的改善作用与阳性对照组相当,并呈现明显的剂量依赖性(小、中、高剂量梯度)。在炎症调控方面,CMP-Fe可抑制血清及结肠促炎因子(IL-6、TNF-α和CRP)的产生,同时提升抑炎因子IL-10及肠道黏膜免疫标志物sIgA的水平。具体而言,CMP-Fe组小鼠结肠组织中IL-6、TNF-α及CRP等促炎因子水平显著低于模型对照组(P < 0.05),提示其可有效调节IDA伴随的炎症反应;尤其在高剂量CMP-Fe干预下,小鼠sIgA水平的恢复效果优于模型对照组(P< 0.05),甚至超过阳性对照组。组织病理学检查显示,CMP-Fe对小鼠的心、肺、脾、肾等器官无明显病理性损伤,表明其具有良好的生物安全性;同时,该螯合物还能显著缓解铁缺乏引起的肠道及肝脏组织病理损伤。综上所述,CMP-Fe可有效改善IDA小鼠的铁代谢紊乱、抑制炎症反应、减轻肠道及肝脏组织损伤,且安全性良好,是一种具有开发潜力的新型有机补铁剂,可用于猫、狗等多种宠物专用粮及营养补充剂的产品开发。
刘蕊莉 , 宋雪盈 , 苗晋鑫 , 赵谋明 , 苏国万 . 鸡肉肽-亚铁螯合物对缺铁性贫血小鼠的影响[J]. 华南理工大学学报(自然科学版), 2026 , 54(2) : 102 -111 . DOI: 10.12141/j.issn.1000-565X.250302
This study systematically investigated the ameliorative effect of Chicken Peptide-Ferrous Chelate (CMP-Fe) on anemia symptoms in mice with iron deficiency anemia (IDA). The intervention effects were comprehensively evaluated through multiple dimensions, including changes in body weight, blood routine parameters, iron metabolism indicators, inflammatory responses, and tissue protection. The experimental results showed that, compared to the model group, all CMP-Fe dose groups (with low-dose, medium-dose, and high-dose administered at Fe 1.0, 2.0 and 3.0 mg/kg, respectively, based on mouse body weight) exhibited significant improvements in body weight, blood routine parameters (including red blood cell count RBC, hemoglobin HGB, hematocrit HCT, mean corpuscular volume MCV, mean corpuscular hemoglobin MCH, mean corpuscular hemoglobin concentration MCHC, and red blood cell distribution width-coefficient of variation RDW-CV), and serum iron metabolism markers (including serum iron SI, total iron-binding TIBC, transferrin receptor TRF, ferritin FER, transferrin saturation TSAT, and unsaturated iron-binding capacity UIBC), with the high-dose group demonstrating the most pronounced effects. The ameliorative effects of CMP-Fe on erythrocyte-related parameters such as RBC, HGB, and HCT were comparable to those of the positive control group and exhibited a clear dose-dependent trend (low, medium, and high dose gradients). In terms of inflammation regulation, CMP-Fe could suppress the production of serum and colonic pro-inflammatory factors (IL-6, TNF-α, and CRP), while elevating the levels of the anti-inflammatory factor IL-10 and the intestinal mucosal immune marker sIgA. Specifically, the levels of pro-inflammatory factors such as IL-6, TNF-α, and CRP in the colonic tissues of mice in the CMP-Fe group were significantly lower than those in the model control group (P < 0.05), suggesting that CMP-Fe effectively modulates the inflammatory response asso-ciated with IDA. Notably, under high-dose CMP-Fe intervention, the recovery of sIgA levels in mice outperformed that of the model control group (P < 0.05) and even exceeded that of the positive control group. Histopathological examination shows that CMP-Fe causes no significant pathological damage to organs such as the heart, lungs, spleen, and kidneys in mice, indicating its favorable biosafety. Meanwhile, this chelate also significantly alleviates pathological injuries in intestinal and liver tissues caused by iron deficiency. In conclusion, CMP-Fe effectively alleviates iron metabolism disorders, inhibits inflammatory responses, and mitigates intestinal and liver tissue damage in mice with IDA, while demonstrating good safety. As a promising novel organic iron supplement, it holds potential for application in the development of specialized pet foods and nutritional supplements for various animals such as cats and dogs.

| [1] | ERRI??E-JERVILD M, KRUU?E C, WEGLEWSKI A,et al .Iron deficiency anemia as a risk factor associated with stroke and cerebral venous thrombosis in children and adults:a systematic review[J].Thrombosis Research,2025,253:109425/1-8. |
| [2] | WICHERT B, SCHADE L, GEBERT S,et al .Energy and protein needs of cats for maintenance,gestation and lactation[J].Journal of Feline Medicine and Surgery,2009,11(10):808-815. |
| [3] | SILVA I C DA, DOS SANTOS P D S, DOS SANTOS JúNIOR O O,et al .Influence of mineral source and inclusion levels of iron,copper,and zinc on the oxidative stability of extruded cat food[J].Animal Feed Science and Technology,2024,315:115997/1-13. |
| [4] | ROUT S R, PRADHAN D, HALDAR J,et al .Recent advances in the formulation strategy to improve iron bioavailability:a review[J].Journal of Drug Delivery Science and Technology,2024,95:105633/1-14. |
| [5] | 丁刘刚,关婷,缪金典,等 .软骨及姜黄联合葛根薏仁提取物对膝骨关节炎大鼠软骨损伤的改善作用[J].华南理工大学学报(自然科学版),2024,52(4):17-25. |
| DING Liugang, GUAN Ting, MIAO Jindian,et al .Improvement effects of co-administration of cartilage extract,turmeric extract,pueraria lobata and coix seed extract on cartilage damage of rats with knee osteoarthritis[J].Journal of South China University of Technology(Natural Science Edition),2024,52(4):17-25. | |
| [6] | LIN S, HU X, YANG X,et al .GLPGPSGEEGKR:Fe2+ chelating characterization and potential transport pathways for improving Fe2+ bioavailability in Caco-2 cells[J].Food Bioscience,2022,48:101806/1-12. |
| [7] | DING X, XU M, LI H,et al .Improvement of in vivo iron bioavailability using mung bean peptide-ferrous chelate[J].Food Research International,2024,190:114602/1-9. |
| [8] | 周雪松,赵谋明,林伟锋,等 .Alcalase酶解鸡肉蛋白及产物的自由基清除活性[J].华南理工大学学报(自然科学版),2006,34(3):117-122. |
| ZHOU Xue-song, ZHAO Mou-ming, LIN Wei-feng,et al .Enzymatic hydrolysis of chicken protein by alcalase and scavenging activity of resulted hydrolysates on free radicals[J].Journal of South China University of Technology (Natural Science Edition),2006,34(3):117-122. | |
| [9] | 吴佳瑄,廖瑞生,况文明,等 .国产鸡肉粉替代鱼粉对大口黑鲈生长性能、肝脏健康及肠道屏障的影响[J].水产学报,2023,47(10):109605/1-11. |
| WU Jiaxuan, LIAO Ruisheng, KUANG Wenming,et al .Effects of replacing fish meal with domestic poultry by-product meal on growth,liver health and intestinal barrier of Micropterus salmoides [J].Journal of Fisheries of China,2023,47(10):109605/1-11. | |
| [10] | CHEN J, YAN Y, ZHANG L,et al .Purification of novel antioxidant peptides from myofibrillar protein hydrolysate of chicken breast and their antioxidant potential in chemical and H2O2-stressed cell systems[J].Food & Function,2021,12(11):4897-4908. |
| [11] | 闫昌誉,丁肇俊,李晓敏,等 .鸡源性活性肽的研究进展[J].今日药学,2022,32(1):4-16. |
| YAN Changyu, DING Zhaojun, LI Xiaomin,et al .Research progress of chicken-derived active peptides[J].Pharmacy Today,2022,32(1):4-16. | |
| [12] | 王彦芝 .菜籽肽亚铁螯合物的制备及特性研究[D].郑州:河南工业大学,2025. |
| [13] | 管玲娟 .阿胶肽铁螯合物调控缺铁性贫血小鼠肠道炎症的作用及其微胶囊化研究[D].无锡:江南大学,2022. |
| [14] | 梅迪鹏 .大豆鲜味肽的制备及呈味机理研究[D].广州:华南理工大学,2024. |
| [15] | SUN N, WANG Z, JIANG H,et al .Angelica sinensis polysaccharides promote extramedullary stress erythropoiesis via ameliorating splenic glycolysis and EPO/STAT5 signaling-regulated macrophages[J].Journal of Molecular Histology,2024,55(5):661-673. |
| [16] | ?ETINKAYA ALTUNTA? S, EVRAN M, GüRKAN E,et al .HbA1c level decreases in iron deficiency anemia[J].Wiener Klinische Wochenschrift,2021,133:102-106. |
| [17] | CANNY S P, OROZCO S L, THULIN N K,et al .Immune mechanisms in inflammatory anemia[J].Annual Review of Immunology,2023,41(1):405-429. |
| [18] | TORSVIK I K, MARKESTAD T, UELAND P M,et al .Evaluating iron status and the risk of anemia in young infants using erythrocyte parameters[J].Pediatric Research,2013,73(2):214-220. |
| [19] | ZHANG H, SUN W, QI J,et al .Antianemic activity,inhibition of oxidative stress,and iron supplementation in mice with iron-deficiency anemia through HG-hawthorn pectin-iron (Ⅲ) complexes[J].ACS Applied Bio Materials,2025,8(3):2419-2428. |
| [20] | YASSIN M A, ALMASRI H A, AL-TIKRITY M A,et al .Neutropenia and lymphocytopenia among arab females with iron deficiency anemia (IDA) and their response to iron therapy[J].Acta Bio-Medica:Atenei Parmensis,2022,93(3):e2022235/1-7. |
| [21] | SABOOR M, ZEHRA A, HAMALI H A,et al .Revisiting iron metabolism,iron homeostasis and iron deficiency anemia[J].Clinical Laboratory,2021,67(3):660-666. |
| [22] | GOSAVI R, JADHAV N B, NASHTE A .Intravenous iron technique evaluation in chronic heart failure with iron deficiency anemia[J].Cureus,2023,15(10):e47778/1-8. |
| [23] | PAGANI A, NAI A, SILVESTRI L,et al .Hepcidin and anemia:a tight relationship[J].Frontiers in Physiology,2019,10:1294/1-7. |
| [24] | GRIGOROPOULOS I, TSIOULOS G, KASTRISSIA-NAKIS A,et al .The safety and potential efficacy of exosomes overexpressing CD24 (EXO-CD24) in mild-moderate COVID-19 related ARDS[J].Respiratory Research,2024,25(1):151/1-13. |
| [25] | HIRANO T .IL-6 in inflammation,autoimmunity and cancer[J].International Immunology,2021,33(3):127-148. |
| [26] | BORGONETTI V, COCETTA V, BIAGI M,et al .Anti-inflammatory activity of a fixed combination of probiotics and herbal extract in an in-vitro model of intestinal inflammation by stimulating Caco-2 cells with LPS-conditioned THP-1 cells medium[J].Minerva Pediatric,2022,74(5):511-518. |
| [27] | WEISS G, GANZ T, GOODNOUGH L T .Anemia of Inflammation[J].Blood,2019,133(1):40-50. |
| [28] | TANOUS O, LEVIN C, SUCHDEV P S,et al .Resolution of iron deficiency following successful eradication of Helicobacter pylori in children[J].Acta Paedia-trica,2022,111(5):1075-1082. |
/
| 〈 |
|
〉 |