

收稿日期: 2024-03-25
网络出版日期: 2024-05-11
基金资助
广东省科技计划项目(2023B110009);季华实验室项目(X210111TD210)
The Effect of Hypoxic Environment on the Enhancement of Collagen Content in Tissue-Engineered Vascular Grafts
Received date: 2024-03-25
Online published: 2024-05-11
Supported by
the Guangdong S & T Programme(2023B110009);the Program of Ji Hua Laboratory(X210111TD210)
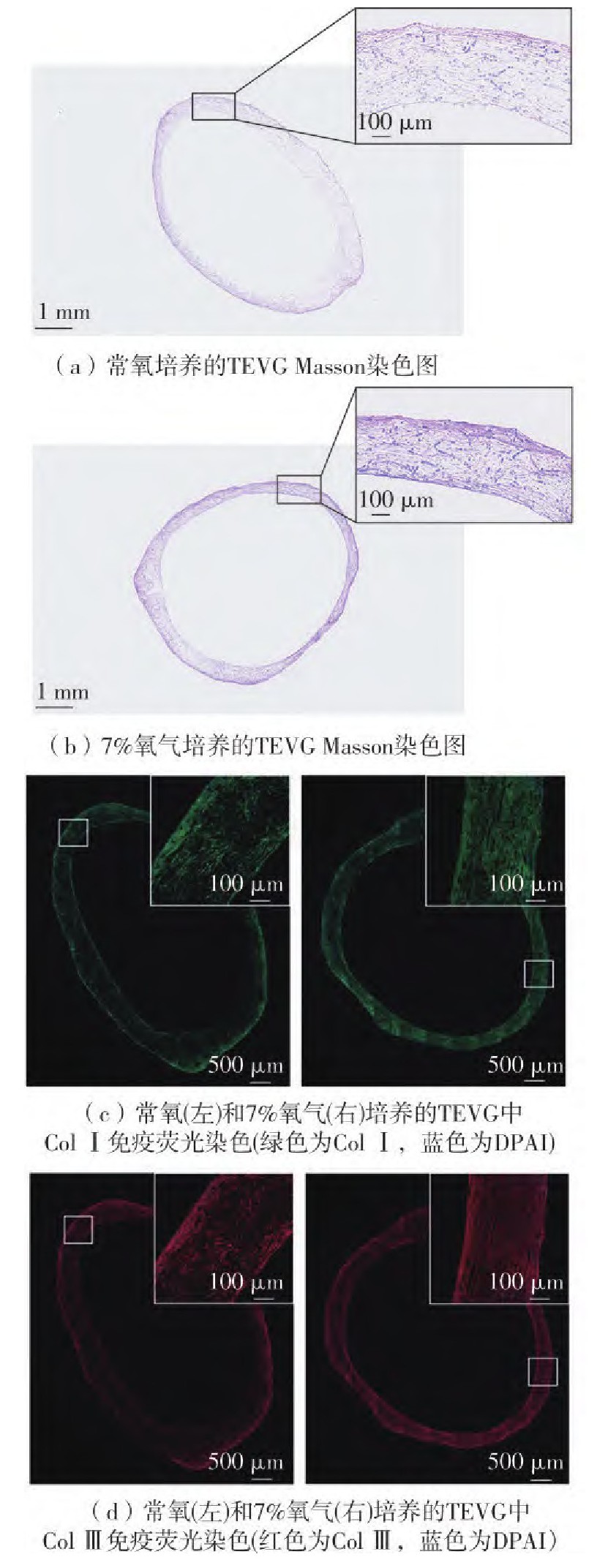
在血管组织工程领域,目前的主要技术方向集中于利用血管平滑肌细胞(VSMCs)在体外增殖和分泌富含胶原蛋白的细胞外基质(ECM),并且施加特定力学刺激重构出具有所需力学性能的生物材料。合适的氧气含量对于体外细胞三维培养至关重要,但在组织工程血管移植物(TEVG)应用方面还缺乏足够研究。为此通过设计不同的氧气含量培养环境,对VSMCs增殖活性检测以确定细胞培养的氧气含量最佳值;进而,在常规氧含量和低氧含量下,对胶原蛋白的基因表达和蛋白分泌情况进行检测,并通过羟脯氨酸检测法对沉积在细胞层中的总胶原含量进行定量;最后,在聚乙醇酸(PGA)支架材料上接种VSMCs进行三维培养形成TEVG,通过组织切片染色和总胶原定量来观察低氧环境下TEVG的培养效果。结果显示:7%氧气条件下VSMCs细胞活性增长最快;低氧环境下VSMCs的Ⅰ型胶原蛋白(Col Ⅰ)和Ⅲ型胶原蛋白(Col Ⅲ)基因表达有所上调,在第5至第9天可以检测到更多胶原蛋白分泌到细胞培养基中,尤其是Col Ⅲ,并且第11天细胞层ECM中总胶原蛋白的含量为常氧组的3.1倍;7%氧气条件可促进PGA支架上VSMCs三维培养过程的胶原沉积,与常氧组相比总胶原含量提高至2.09倍,并形成更致密的胶原纤维。结果表明,选择低氧环境可以促进组织工程血管移植物胶原蛋白的含量,可为进一步优化TEVG的体外培养条件提供依据。
林展翼 , 肖聪 , 许健宜 , 刘青 , 孙盱衡 , 方丽君 . 低氧环境对组织工程血管移植物胶原蛋白含量的提升作用[J]. 华南理工大学学报(自然科学版), 2025 , 53(1) : 129 -135 . DOI: 10.12141/j.issn.1000-565X.240137
In the field of vascular tissue engineering, current technological advancements are focused on the proli-feration and secretion of collagen-rich extracellular matrix (ECM) by vascular smooth muscle cells (VSMCs) in vitro, coupled with specific mechanical stimuli to reconstruct biomaterials with desired mechanical properties. The optimal oxygen concentration is crucial for three-dimensional cell culture in vitro. However, there is a lack of sufficient research regarding its application in tissue-engineered vascular graft (TEVG). To this end, this study designed varying oxygen concentration environments to determine the optimal value for VSMCs culture by assessing cell proliferation activity. Furthermore, collagen gene expression and protein secretion were measured under normoxic and hypoxic conditions, along with quantification of total collagen content deposited in the cell layer using hydroxyproline assay. Subsequently, VSMCs were seeded on polyglycolic acid (PGA) scaffolds for three-dimensional culturing to form TEVG, and the effects of hypoxic conditions on TEVG cultivation were observed through histological staining and total collagen quantification. The results indicate that VSMC cell activity increased most rapidly at 7% oxygen concentration; under hypoxic conditions, an upregulation of type I (Col I) and type Ⅲ (Col Ⅲ) collagen gene expression was observed, with an increased secretion of collagen into the culture medium between days 5 and 9, particularly Col Ⅲ. Furthermore, the total collagen content on the 11th day demonstrates a 3.1-fold increase relative to the normoxic group. The 7% oxygen concentration facilitated collagen deposition during the three-dimensional culture of VSMCs on PGA scaffolds, resulting in a 2.09-fold increase in total collagen content compared to the normoxic group. Additionally, the formation of more dense collagen fibrils was observed. These findings indicate that the utilization of a hypoxic environment can enhance the collagen content in TEVG, thereby providing a foundation for the further optimization of in vitro culture conditions for TEVG.

| 1 | TSAO C W, ADAY A W, ALMARZOOQ Z I,et al .Heart disease and stroke statistics-2022 update:a report from the American Heart Association[J].Circulation,2022,145(8):e153-e639. |
| 2 | World Health Organization .A global brief on hypertension[R].Geneva:World Health Organization,2013. |
| 3 | LLOYD-JONES D, ADAMS R, CARNETHON M, et al .Heart disease and stroke statistics—2009 update:a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee[J].Circulation,2009,119(3):e21-e181. |
| 4 | HUANG Y, GLOVICZKI P, NOEL A A,et al .Early complications and long-term outcome after open surgical treatment of popliteal artery aneurysms:is exclusion with saphenous vein bypass still the gold standard?[J].Journal of Vascular Surgery,2007,45(4):706 -713. |
| 5 | SWAIN T W, CALLIGARO K D, DOUGHERTY M D .Management of infected aortic prosthetic grafts[J].Vascular and Endovascular Surgery,2004,38(1):75-82. |
| 6 | GEELHOED W J, MORONI L, ROTMANS J I .Utili-zing the foreign body response to grow tissue engineered blood vessels in vivo[J].Journal of Cardiovascular Translational Research,2017,10:167-179. |
| 7 | NIKLASON L E, GAO J, ABBOTT W M,et al .Functional arteries grown in vitro[J].Science,1999,284(5413):489-493. |
| 8 | NAEGELI K M, KURAL M H, LI Y,et al .Bioengineering human tissues and the future of vascular replacement[J].Circulation Research,2022,131(1):109-126. |
| 9 | WANG H W, CHENG C W, LI C W,et al .Fabrication of pillared PLGA microvessel scaffold using femtosecond laser ablation[J].International Journal of Nanomedicine,2012,7:1865-1873. |
| 10 | PENNINGS I, van HAAFTEN E E, JUNGST T,et al .Layer-specific cell differentiation in bi-layered vascular grafts under flow perfusion[J].Biofabrication,2019,12(1):015009/1-13. |
| 11 | MCFETRIDGE P S,ABE K, HORROCKS M,et al .Vascular tissue engineering:bioreactor design considerations for extended culture of primary human vascular smooth muscle cells[J].ASAIO Journal,2007,53(5):623-630. |
| 12 | TSCHOEKE B, FLANAGAN T C, KOCH S,et al .Tissue-engineered small-caliber vascular graft based on a novel biodegradable composite fibrin-polylactide scaffold[J].Tissue Engineering Part A,2009,15(8):1909-1918. |
| 13 | FATHOLLAHIPOUR S, PATIL P S, LEIPZIG N D .Oxygen regulation in development:lessons from embryogenesis towards tissue engineering[J].Cells Tissues Organs,2019,205(5/6):350-371. |
| 14 | van VLIMMEREN M A A, DRIESSEN-MOL A, van DEN BROEK M,et al .Controlling matrix formation and cross-linking by hypoxia in cardiovascular tissue engineering[J]?.Journal of Applied Physiology,2010,109(5):1483-1491. |
| 15 | SHIMODA L A .Cellular pathways promoting pulmonary vascular remodeling by hypoxia[J].Physiology,2020,35(4):222-233. |
| 16 | FRANTZ C, STEWART K M, WEAVER V M .The extracellular matrix at a glance[J].Journal of Cell Science,2010,123(24):4195-4200. |
| 17 | von der MARK K .Localization of collagen types in tissues[J].International Review of Connective Tissue Research,1981,9:265-324. |
| 18 | NIKLASON L E, LAWSON J H .Bioengineered human blood vessels[J].Science,2020,370(6513):eaaw8682/1-9. |
| 19 | DELLA ROCCA Y, FONTICOLI L, RAJAN T S,et al .Hypoxia:molecular pathophysiological mechanisms in human diseases[J].Journal of Physiology and Biochemistry,2022,78(4):739-752. |
| 20 | FANG L, MEI J, YAO B,et al .Hypoxia facilitates proliferation of smooth muscle cells derived from pluripotent stem cells for vascular tissue engineering[J].Journal of Tissue Engineering and Regenerative Medicine,2022,16(8):744-756. |
| 21 | HU C, HUANG S, WU F,et al .MicroRNA?219?5p participates in cyanotic congenital heart disease progression by regulating cardiomyocyte apoptosis[J].Experimental and Therapeutic Medicine,2021,21:36/1-7. |
| 22 | YANG G, LIN C .Long noncoding RNA SOX2‐OT exacerbates hypoxia‐induced cardiomyocytes injury by regulating miR‐27a‐3p/TGFβR1 axis[J].Cardiovascular Therapeutics,2020,2020(1):2016259/1-11. |
| 23 | BROWER G L, GARDNER J D, FORMAN M F, et al .The relationship between myocardial extracellular matrix remodeling and ventricular function[J].European Journal of Cardio-Thoracic Surgery,2006,30(4):604-610. |
| 24 | MIENALTOWSKI M J, BIRK D E .Structure,physiology,and biochemistry of collagens[J].Advances in Experimental Medicine and Biology,2013,802:5-29. |
| 25 | TOBA H, CANNON P L, YABLUCHANSKIY A,et al .Transgenic overexpression of macrophage matrix metalloproteinase-9 exacerbates age-related cardiac hypertrophy,vessel rarefaction,inflammation,and fibrosis[J].American Journal of Physiology-Heart and Circulatory Physiology,2017,312(3):H375-H383. |
| 26 | LIU X, WU H, BYRNE M,et al .Type III collagen is crucial for collagen I fibrillogenesis and for normal cardiovascular?development[J].Proceedings of the National Academy of Sciences of the United States of America,1997,94(5):1852-1856. |
| 27 | FLEISCHMAJER R, MACDONALD E D, PERLISH J S,et al .Dermal collagen fibrils are hybrids of type I and type III collagen molecules[J].Journal of Structural Biology,1990,105(1/2/3):162-169. |
| 28 | RONG N, MISTRIOTIS P, WANG X,et al .Restoring extracellular matrix synthesis in senescent stem cells[J].The FASEB Journal,2019,33(10):10954-10965. |
| 29 | ERYILMAZ E, TEIZER W, HWANG W .In vitro analysis of the co-assembly of type-I and type-III collagen[J]?.Cellular and Molecular Bioengineering,2017,10:41-53. |
| 30 | GAO J, GUO Z, ZHANG Y,et al .Age-related changes in the ratio of Type I/III collagen and fibril diameter in mouse skin[J].Regenerative Biomaterials,2023,10:rbac110/1-9. |
/
| 〈 |
|
〉 |