

收稿日期: 2022-05-31
网络出版日期: 2022-08-26
基金资助
国家自然科学基金资助项目(51672086);广东省自然科学基金资助项目(2017B030308005)
Electrochemical Performance of Rb-Doped Na1.25V3O8 Nanorods as Cathode for Zinc-Ion Batteries
Received date: 2022-05-31
Online published: 2022-08-26
Supported by
the National Natural Science Foundation of China(51672086);the Natural Science Foundation of Guangdong Province(2017B030308005)
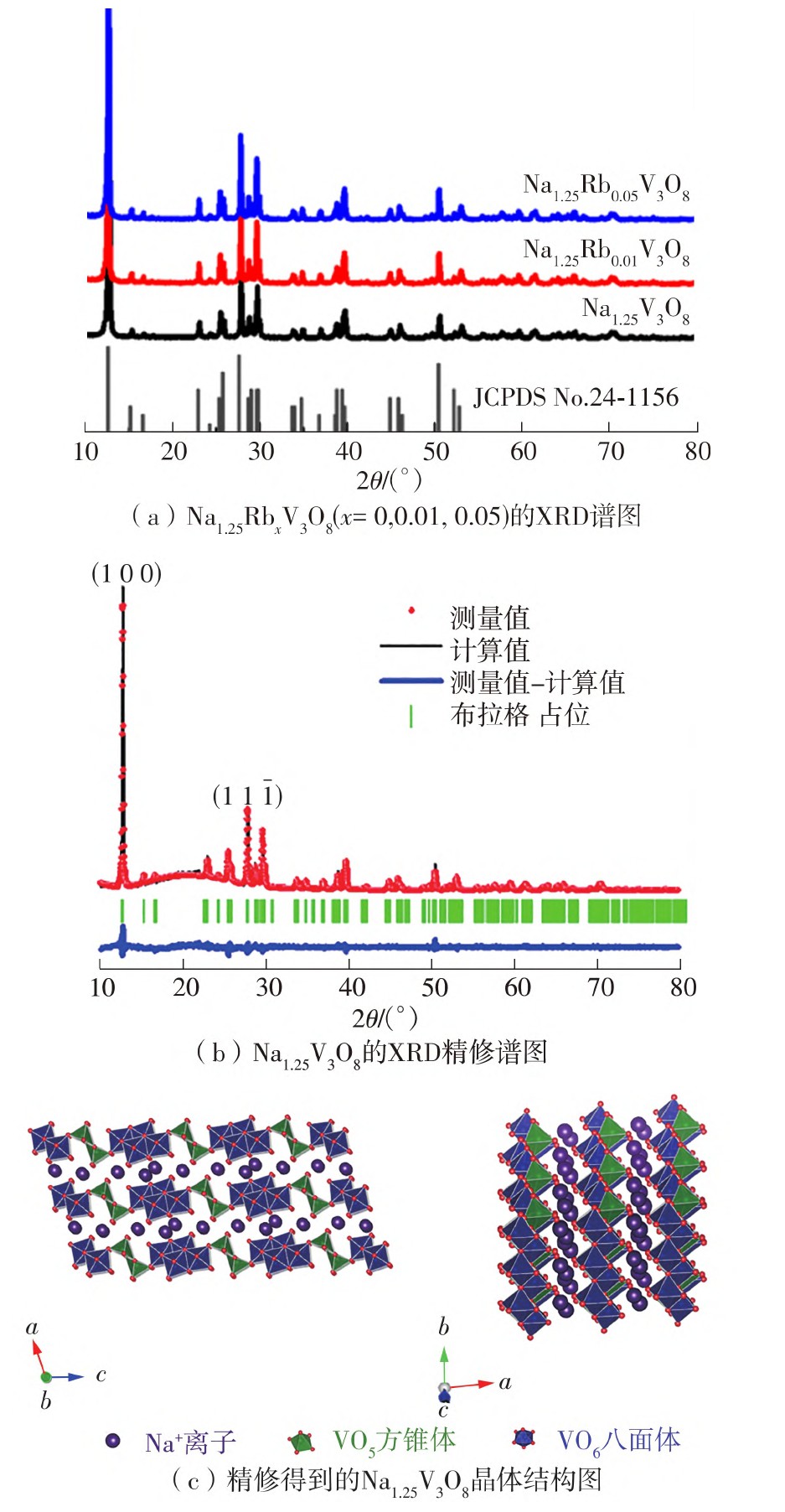
为了探究作为锌离子电池正极的Na1.25V3O8的电化学性能是否还具有进一步提升的空间,文中通过水热-固相法成功合成了纯Na1.25V3O8和铷掺杂Na1.25Rb x V3O8纳米棒。根据XRD谱图可以看出,随着铷含量的增加衍射峰向低角度偏移且没有新的衍射峰生成,表明Na1.25Rb x V3O8样品为纯相且保持空间群为P21/m的结构对称性。这一结果说明铷离子成功进入了Na1.25Rb x V3O8层状化合物的层间。SEM和TEM形貌分析结果显示,随着Rb+掺杂量的增加,纳米棒的形貌变得更大、更厚,会降低材料的比表面积,导致阻抗的增加和Zn2+离子扩散系数的降低,对电化学性能产生不利的影响。EIS和GITT测试结果也证实,Rb+的掺入会使其比容量降低。电化学测试结果显示,虽然未掺杂的Na1.25V3O8纳米棒表现出370 mA·h/g的初始比容量,但随着充放电过程的进行其比容量衰减很快,在电流密度为100 mA/g条件下长循环100圈后的放电比容量衰减了初始容量的50%;而Rb+掺杂的纳米棒尽管比容量有所降低,但其循环稳定性和倍率性能均得到显著提升。其中Rb掺杂量为1%的样品初始比容量为336 mA·h/g,在相同条件下循环100圈后和初始比容量相比只衰减了6%;当Rb掺杂量增加到5%时,尽管初始比容量只有217 mA·h/g,但100圈后放电比容量和初始比容量相比几乎没有衰减。利用XPS能谱和原位XRD技术对其锌离子存储机理进行了深入分析,该水系锌离子电池体系中Na1.25Rb x V3O8正极材料的氧化还原反应机理主要以化学转化反应为主,其中只有充电反应过程是可逆的。
关键词: 锌离子电池; 正极材料; Na1.25V3O8; 铷掺杂; 纳米棒
赵彦明, 廖锦辉 . 铷掺杂Na1.25V3O8纳米棒作为锌离子电池正极材料的电化学性能[J]. 华南理工大学学报(自然科学版), 2023 , 51(3) : 63 -73 . DOI: 10.12141/j.issn.1000-565X.220326
In order to know whether there is space for the improvement of the electrochemical performance of Na1.25V3O8 as cathode for zinc-ion batteries, this study successfully synthesized pure Na1.25V3O8 and Rb+ ion doped Na1.25V3O8 nanorods through hydrothermal method and solid phase method. With the help of XRD patterns, the diffraction peak shifts to a low angle and no new diffraction peak is generated with the increase of rubidium content, indicating that the Na1.25Rb x V3O8 sample is pure phase and maintain the structural symmetry of P21/m space group. This result indicate that rubidium ions have successfully entered the interlayer of Na1.25Rb x V3O8 layered compound. The results of SEM images and TEM images analysis show that, with the insertion of Rb+, scale of Na1.25V3O8 nanorods become thicker and larger, which would reduce the specific surface and lead to the increase of impedance and reducing the ion diffusion coefficients, resulting in poor electrochemical performance. EIS and GITT results further confirm that the insertion of Rb+ will lead to the decrease in capacity. Electrochemical testing results show that bare Na1.25V3O8 nanorods exhibit about 370 mA·h/g initial capacity, but it decreases fast with the increasing times of charge and discharge circle. After 100 cycles of discharge and charge processes at current density of 100 mA/g, discharge capacity decays about 50% compared to initial capacity. Although the specific capacity of Rb+-doped nanorods is reduced, their cycle stability and rate performance are significantly improved. When the amount of doped Rb is 1%, the initial capacity is 336 mA·h/g, but after 100 cycles of discharge and charge processes under the same conditions, the discharge capacity only decays about 6% compared to initial discharge capacity. When the amount of Rb increases to 5%, the initial capacity is merely 217 mA·h/g, but no capacity lost compared to initial capacity. The mechanism of zinc ion storage was analyzed through in-situ XRD and ex-situ XPS. The redox reaction mechanism of Na1.25Rb x V3O8 cathode material in the ZIBs is mainly based on chemical conversion reaction, in which only the charging reaction process is reversible.

Key words: zinc-ion batteries; cathode material; Na1.25V3O8; Rb-doped; nanorods
| 1 | WHITTINGHAM M S .Introduction:batteries[J]?.Chemical Reviews,2014,114(23):11413. |
| 2 | YAN M Y, HE P, CHEN Y,et al .Water-lubricated intercalation in V2O5·nH2O for high-capacity and high-rate aqueous rechargeable zinc batteries[J].Advanced Materials,2018,30(1):1703725-1-6. |
| 3 | ZHU K Y, WU T, HUANG K V .NaCa0.6V6O16·3H2O as an ultra-stable cathode for Zn-ion batteries:The roles of pre-inserted dual-cations and structural water in V3O8 layer[J].Advanced Energy Materials,2019,9(38):190 1968-1-12. |
| 4 | MUENCH S, WILD A, FRIEBE C,et al .Polymer-based organic batteries[J].Chemical Reviews,2016,116(16):9438-9484. |
| 5 | AKINYELE D O, RAYUDU R K .Review of energy storage technologies for sustainable power networks[J].Sustainable Energy Technologies and Assessments,2014,8:74-91. |
| 6 | HAMEER S, VAN NIEKERK J L .A review of large-scale electrical energy storage[J].International Journal of Energy Research,2015,39(9):1179-1195. |
| 7 | GOODENOUGH J B, KIM Y .Challenges for rechargeable Li batteries[J].Chemistry of Materials,2010,22(3):587-603. |
| 8 | HU P, ZHU T, WANG X P,et al .Highly durable Na2V6O16·1.63H2O nanowire cathode for aqueous zinc-ion battery[J].Nano Letters,2018,18(3):1758-1763. |
| 9 | PAN H L, SHAO Y Y, YAN P F,et al .Reversible aqueous zinc/manganese oxide energy storage from conversion reactions[J].Nature Energy,2016,1(5):1-17. |
| 10 | XU C J, LI B H, DU H D,et al .Energetic zinc ion chemistry:The rechargeable zinc ion battery[J].Angewandte Chemie,2012,124(4):957-959. |
| 11 | ALFARUQI M H, MATHEW V,GIM J,et al .Electrochemically induced structural transformation in a γ- MnO2 cathode of a high capacity zinc-ion battery system[J].Chemistry of Materials,2015,27(10):3609-3620. |
| 12 | ZHANG N, CHENG F Y, LIU Y C,et al .Cation-deficient spinel ZnMn2O4 cathode in Zn(CF3SO3)2 electrolyte for rechargeable aqueous Zn-ion battery[J].Journal of the American Chemical Society,2016,138(39):12894-12901. |
| 13 | ZHANG B H, LIU Y, WU X W,et al .An aqueous rechargeable battery based on zinc anode and Na0.95MnO2 [J].Chemical Communications,2014,50(10):1209-1211. |
| 14 | LIU Z, PULLETIKURTHI G, ENDRES F .A Prussian blue/zinc secondary battery with a bio-ionic liquid-water mixture as electrolyte[J].ACS Applied Materials & Interfaces,2016,8(19):12158-12164. |
| 15 | ZHANG L Y, CHEN L, ZHOU X F,et al .Morphology-dependent electrochemical performance of zinc hexacyanoferrate cathode for zinc-ion battery[J].Scientific Reports,2015,5(1):18263. |
| 16 | HOU Z G, ZHANG X Q, LI X N,et al .Surfactant widens the electrochemical window of an aqueous electrolyte for better rechargeable aqueous sodium/zinc battery[J].Journal of Materials Chemistry A,2017,5(2):730-738. |
| 17 | ZHANG L Y, CHEN L, ZHOU X F,et al .Towards high-voltage aqueous metal-ion batteries beyond 1.5 V:The zinc/zinc hexacyanoferrate system[J].Advanced Energy Materials,2015,5(2):1400930-1-5. |
| 18 | MING J, GUO J, XIA C,et al .Zinc-ion batteries:Materials,mechanisms,and applications[J].Materials Science & Engineering,2019,135:58-84. |
| 19 | 衡永丽,谷振一,郭晋芝,等 .水系锌离子电池用钒基正极材料的研究进展[J].物理化学学报,2021,37(3):2005013-1-16. |
| HENG Yong-li, GU Zhen-yi, GUO Jin-zhi,et al .Research progresses on vanadium-based cathode materials for aqueous zinc-ion batteries[J].Acta Physico-Chimica Sinica,2021,37(3):2005013-1-16. | |
| 20 | HE H N, JIN G H, WANG H Y,et al .Annealed NaV3O8 nanowires with good cycling stability as a novel cathode for Na-ion batteries[J].Journal of Materials Chemistry A,2014,2(10):3563-3570. |
| 21 | CAO Y H, FANG D, WANG C,et al .Novel aligned sodium vanadate nanowire arrays for high-performance lithium-ion battery electrodes[J].RSC Advances,2015,5(53):42955-42960. |
| 22 | CAO Y H, WANG J Y, CHEN X T,et al .Nanostructured sodium vanadate arrays as an advanced cathode material in high-performance sodium-ion batteries[J].Materials Letters,2019,237:122-125. |
| 23 | KIM S J, TANG C R, SINGH G,et al .New insights into the reaction mechanism of sodium vanadate for an aqueous Zn ion battery[J].Chemistry of Materials,2020,32(5):2053-2060. |
| 24 | HE P, ZHANG G B, LIAO X B,et al .Sodium ion stabilized vanadium oxide nanowire cathode for high-performance zinc-ion batteries[J].Advanced Energy Materials,2018,8(10):1702463-1-6. |
| 25 | DONG Y F, LI S, ZHAO K N,et al .Hierarchical zigzag Na1.25V3O8 nanowires with topotactically encoded superior performance for sodium-ion battery cathodes[J].Energy & Environmental Science,2015,8(6):1267-1275. |
| 26 | WANG J, POLLEUX J,LIM J,et al .Pseudocapacitive contributions to electrochemical energy storage in TiO2 (anatase) nanoparticles[J].Journal of Physical Chemistry C,2007,111(140):14925-14931. |
| 27 | SIMON P, GOGOTSI Y, DUNN B .Where do batteries end and supercapacitors begin[J].Science,2014,343(6176):1210-1211. |
| 28 | CONWAY B E, BIRSS V, WOJTOWICZ J .The role and utilization of pseudocapacitance for energy storage by supercapacitors[J].Journal of Power Sources,1997,66(1/2):1-14. |
| 29 | LINDATROM H, SODERGREN S, SOLBRAND A,et al .Li+ ion insertion in TiO2 (anatase).2.Voltammetry on nanoporous films[J].Journal of Physical Chemistry B,1997,101(39):7717-7722. |
| 30 | SOUNDHARRAJAN V, SAMBANDAM B, KIM S,et al .Na2V6O16·3H2O barnesite nanorod:An open door to display a stable and high energy for aqueous rechargeable Zn-ion batteries as cathodes[J].Nano Letters,2018,18(4):2402-2410. |
| 31 | GUO X, FANG G Z, ZHANG W Y,et al .Mechanistic insights of Zn2+ storage in sodium vanadates[J].Advanced Energy Materials,2018,8(27):1801819-1-7. |
/
| 〈 |
|
〉 |