

收稿日期: 2022-03-14
网络出版日期: 2022-04-06
基金资助
广东省自然科学基金资助项目(2021A1515012110)
Antitumor Activity of Ferulic Acid and Its Colonic Metabolites
Received date: 2022-03-14
Online published: 2022-04-06
Supported by
the Natural Science Foundation of Guangdong Province(2021A1515012110)
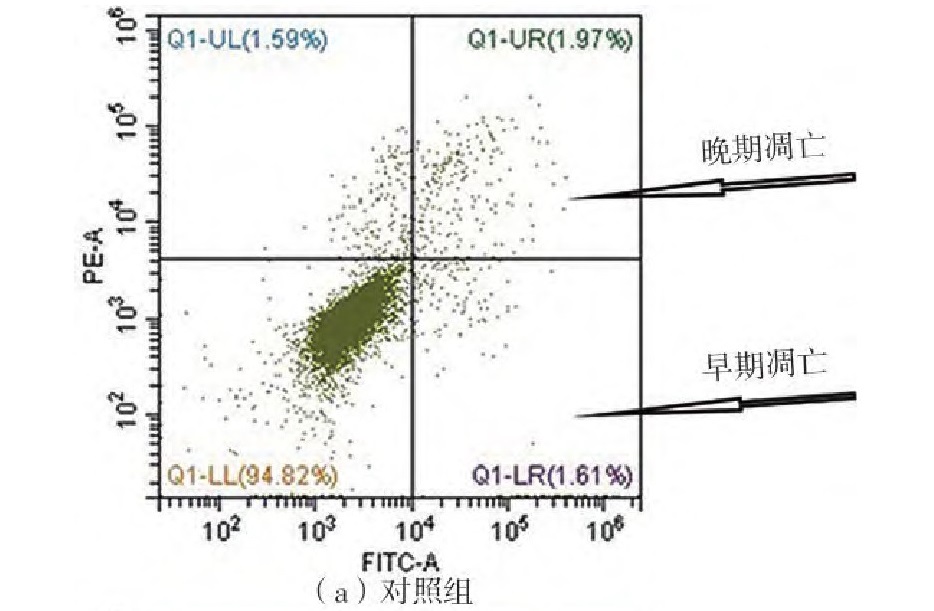
阿魏酸(FA)是一种常见的膳食多酚,广泛存在于植物组织中,具有抗氧化、抗炎、抗血栓、降血糖等多种生物活性。由于膳食多酚的生物活性很大程度上取决于它们在体内的消化吸收,近年来,有关膳食多酚肠道代谢物的研究逐渐引起了人们的兴趣。阿魏酸无法在胃和小肠中被吸收,但在结肠酯酶等的作用下,能够转化为一系列羟基苯丙酸化合物。文中选取了阿魏酸在结肠中的3种主要代谢物——3,4-二羟基苯丙酸(3,4diOHPPA)、3-(3-羟基苯基)丙酸(3OHPPA)和3-苯丙酸3PPA),研究了它们的抗氧化和抗肿瘤活性。结果表明,代谢物3,4diOHPPA的抗氧化活性优于FA和代谢物3OHPPA、3PPA。阿魏酸及其3种代谢物能够显著抑制人肝癌细胞HepG2的增殖,它们的半最大效应浓度(EC50值)分别为1.82 mmol/L(FA)、0.74 mmol/L(3,4diOHPPA)、7.77 mmol/L(3PPA)和4.52 mmol/L(3PPA)。细胞周期实验表明,FA及其3种代谢物能够有序调控HepG2细胞周期进程,将细胞周期阻滞于G2期或S期。FA及其3种代谢物也能够剂量依赖性地诱导HepG2细胞凋亡,其中,FA和3,4diOHPPA能够分别将HepG2细胞总凋亡率提高至15.47%和71.84%(对照组为4.23%)。经过24 h预处理后,FA通过上调Bax、p53基因,下调CDK-2、CDK-4基因发挥抗增殖效果;3,4diOHPPA则通过上调Bax、caspase-3基因,下调CDK-2、CDK-4基因抑制HepG2增殖。研究结果表明代谢物3,4diOHPPA的抗氧化和抗HepG2细胞增殖能力优于FA,为FA及其代谢物的抗肿瘤活性研究提供了理论支持,揭示了FA肠道代谢物的健康益处。
郑必胜, 杨文涵, 徐秋雄, 等 . 阿魏酸及其结肠代谢物的抗肿瘤活性[J]. 华南理工大学学报(自然科学版), 2022 , 50(8) : 30 -40 . DOI: 10.12141/j.issn.1000-565X.220113
Ferulic acid (FA) is a common dietary polyphenol which is widely found in plant tissues. It has various biological activities such as antioxidant, anti-inflammatory, antithrombotic, and hypoglycemic. Due to the biological activities of dietary polyphenols largely depend on their digestion and absorption in vivo, studies on the intestinal metabolites of dietary polyphenols have gradually attracted researchers’ interest in recent years. Ferulic acid cannot be absorbed in the stomach and small intestine, but it can be converted into a series of hydroxyphenylpropionic acid compounds under the action of colonic esterases. This study selected three main colonic metabolites of FA, namely, 3-(3,4-dihydroxyphenyl) propionic acid (3,4diOHPPA), 3-(3-hydroxyphenyl) propionic acid (3OHPPA) and 3-phenylpropionic acid (PPA), to evaluate their antioxidant and antitumor activities. The results show that the antioxidant activity of metabolite 3,4diOHPPA is superior to that of FA and metabolites 3OHPPA and 3PPA. FA and its three metabolites can significantly inhibit the proliferation of HepG2 with EC50 values of 1.82 mmol/L (FA), 0.74 mmol/L (3,4diOHPPA), 7.77 mmol/L (3PPA) and 4.52 mmol/L (3PPA), respectively. Cell-cycle experiments show that FA and its three metabolites can regulate HepG2 cell cycle progression in an orderly manner, blocking the cell cycle in G2 or S phase. FA and its three metabolites can also induce apoptosis of HepG2 in a dose-dependent manner. Among them, FA and 3,4diOHPPA can increase the total apoptosis rate of HepG2 cells to 15.47% and 71.84% (4.23% for the control). After a pretreatment for 24 h, FA exerts its antiproliferative effects by upregulating Bax, p53 genes and downregulating CDK-2, CDK-4 genes; 3,4diOHPPA inhibits the proliferation of HepG2 by upregulating Bax, caspase-3 genes and downregulating CDK-2, CDK-4 genes. All these findings show that the anti-oxidative and anti-HepG2 cell proliferation ability of the metabolite 3,4diOHPPA is superior to that of FA. This research provides theoretical support for the antitumor activity of FA and its colonic metabolites, revealing the health benefits of FA intestinal metabolites.

Key words: ferulic acid; colonic metabolites; antitumor activity; proliferation
| 1 | BARONE E, CALABRESE V, MANCUSO C .Ferulic acid and its therapeutic potential as a hormetin for age-related diseases[J].Biogerontology,2009,10(2):97-108. |
| 2 | CHOWDHURY S, GHOSH S, DAS A K,et al .Ferulic acid protects hyperglycemia-induced kidney damage by regulating oxidative insult,inflammation and autophagy[J].Frontiers in Pharmacology,2019,10:3389. |
| 3 | GIZAWY H A EL, BOSHRA S A, MOSTAFA A,et al . Pimenta dioica (L.) Merr. bioactive constituents exert anti-SARS-CoV-2 and anti-inflammatory activities: molecular docking and dynamics,in vitro,and in vivo studies[J].Molecules,2021,26(19):5844. |
| 4 | CHOI J H, PARK J K, KIM K M,et al .In vitro and in vivo antithrombotic and cytotoxicity effects of ferulic acid[J].Journal of Biochemical and Molecular Toxicology,2018,32(1):1002. |
| 5 | JUNG E H, KIM S R, HWANG I K,et al .Hypoglycemic effects of a phenolic acid fraction of rice bran and ferulic acid in C57BL/KsJ-db/db mice[J].Journal of Agricultural and Food Chemistry,2007,55(24):9800-9804. |
| 6 | MANCUSO C, SANTANGELO R .Ferulic acid: pharmacological and toxicological aspects[J].Food and Chemical Toxicology,2014,65:185-195. |
| 7 | LI D, RUI Y X, GUO S D,et al .Ferulic acid: a review of its pharmacology,pharmacokinetics and derivatives[J].Life Sci,2021,284:119921. |
| 8 | RECHNER A R, KUHNLE G, BREMNER P,et al .The metabolic fate of dietary polyphenols in humans[J].Free Radical Bio Med,2002,33(2):220-235. |
| 9 | CHESSON A, PROVAN G J, RUSSELL W R,et al .Hydroxycinnamic acids in the digestive tract of livestock and humans[J].Journal of the Science of Food and Agriculture,1999,79(3):373-378. |
| 10 | ZHAO Z H, MOGHADASIAN M H .Chemistry,natural sources,dietary intake and pharmacokinetic properties of ferulic acid:a review[J].Food Chem,2008,109(4):691-702. |
| 11 | BRAUNE A, BUNZEL M, YONEKURA R,et al .Conversion of dehydrodiferulic acids by human intestinal microbiota[J].Journal of Agricultural and Food Chemistry,2009,57(8):3356-3362. |
| 12 | ANSON N M, SELINHEIMO E, HAVENAAR R,et al.Bioprocessing of wheat bran improves in vitro bioaccessibility and colonic metabolism of phenolic compounds[J]. Journal of Agricultural and Food Chemistry,2009,57(14):6148-6155. |
| 13 | RUSSELL W R, SCOBBIE L, CHESSON A,et al .Anti-inflammatory implications of the microbial transformation of dietary phenolic compounds[J].Nutrition and Cancer-An International Journal,2008,60(5):636-642. |
| 14 | CELIK E E, GOKMEN V, FOGLIANO V .Soluble Antioxidant compounds regenerate the antioxidants bound to insoluble parts of foods[J].Journal of Agricultural and Food Chemistry,2013,61(43):10329-10334. |
| 15 | ZHENG S L, ZHANG H, LIU R H,et al .Do short chain fatty acids and phenolic metabolites of the gut have synergistic anti-inflammatory effects? - New insights from a TNF-alpha-induced Caco-2 cell model[J].Food Res Int,2021,139:109833/1-13. |
| 16 | ADOM K K, LIU R H .Rapid peroxyl radical scavenging capacity (PSC) assay for assessing both hydrophilic and lipophilic antioxidants[J].Journal of Agricultural and Food Chemistry,2005,53(17):6572-6580. |
| 17 | WOLFE K L, LIU R H .Cellular antioxidant activity (CAA) assay for assessing antioxidants,foods,and dietary supplements[J]. Journal of Agricultural and Food Chemistry,2007,55(22):8896-8907. |
| 18 | FELICE D L, SUN J, LIU R H .A modified methylene blue assay for accurate cell counting[J].Journal of Functional Foods,2009,1(1):109-118. |
| 19 | BI H C, KRAUSZ K W, MANNA S K,et al .Optimization of harvesting,extraction,and analytical protocols for UPLC-ESI-MS-based metabolomic analysis of adherent mammalian cancer cells[J].Anal Bioanal Chem,2013,405(15):5279-5289. |
| 20 | ZHANG X W, ZHANG M W, DONG L H,et al .Phytochemical profile,bioactivity,and prebiotic potential of bound phenolics released from rice bran dietary fiber during in vitro gastrointestinal digestion and colonic fermentation[J].Journal of Agricultural and Food Chemistry,2019,67(46):12796-12805. |
| 21 | KIKUZAKI H, HISAMOTO M, HIROSE K,et al .Antioxidant properties of ferulic acid and its related compounds[J].Journal of Agricultural and Food Chemistry,2002,50(7):2161-2168. |
| 22 | GRAF E .Antioxidant potential of ferulic acid[J].Free Radic Biol Med,1992,13(4):435-448. |
| 23 | AFSHARI A R, MOLLAZADEH H, HENNEY N C,et al .Effects of statins on brain tumors:a review[J].Semin Cancer Biol,2021,73:116-133. |
| 24 | D’ARCY M S .Cell death:a review of the major forms of apoptosis,necrosis and autophagy[J].Cell Biol Int,2019,43(6):582-592. |
| 25 | ELMORE S .Apoptosis:a review of programmed cell death[J].Toxicol Pathol,2007,35(4):495-516. |
| 26 | 董雅洁,高维娟 .bcl-2、bax、caspase-3在细胞凋亡中的作用及其关系[J].中国老年学杂志,2012,32(21):4828-4830. |
| 26 | DONG Yajie, GAO Weijuan .Effect of bcl-2,bax and caspase-3 on cell apoptosis and corresponding relationship[J].Chinese Journal of Gerontology,2012,32(21):4828-4830. |
| 27 | WAJANT H, PFIZENMAIER K, SCHEURICH P .Tumor necrosis factor signaling[J].Cell Death Differ,2003,10(1):45-65. |
| 28 | OTTO T, SICINSKI P .Cell cycle proteins as promising targets in cancer therapy[J].Nat Rev Cancer,2017,17(2):93-115. |
| 29 | JIN Y, FAN Y, YAN E Z,et al .Effects of sodium ferulate on amyloid-beta-induced MKK3/MKK6-p38 MAPK-Hsp27 signal pathway and apoptosis in rat hippocampus[J].Acta Pharmacol Sin,2006,27(10):1309-1316. |
/
| 〈 |
|
〉 |